A copper-nickel diffusion couple similar to that shown in Figure 5.1a is fashioned. After a 700-h heat
Question:
A copper-nickel diffusion couple similar to that shown in Figure 5.1a is fashioned. After a 700-h heat treatment at 1100?C (1373 K) the concentration of Cu is 2.5 wt% at the 3.0-mm position within the nickel. At what temperature must the diffusion couple need to be heated to produce this same concentration (i.e., 2.5 wt% Cu) at a 2.0-mm position after 700 h? The preexponential and activation energy for the diffusion of Cu in Ni are given in Table 5.2.
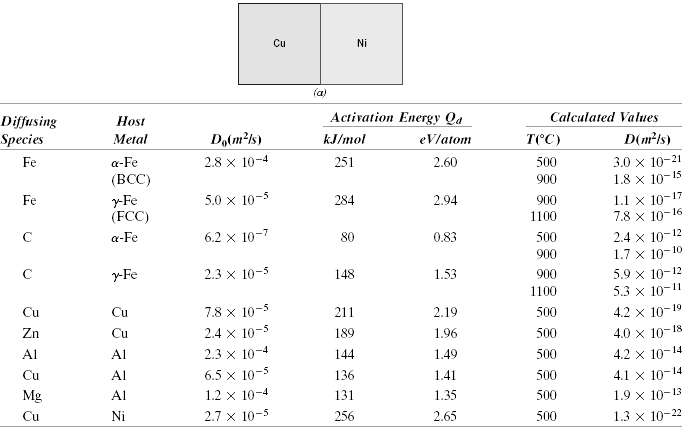
Transcribed Image Text:
Cu Ni (a) Activation Energy Qa Calculated Values Host Diffusing Species Dalm?1s) D(m²ls) тес) Metal kJ/mol eVlatom 3.0 x 10-21 1.8 x 10-15 2.8 x 10-4 Fe a-Fe 251 2.60 500 (BCC) 900 1.1 x 10-17 7.8 x 10-16 y-Fe (FCC) 5.0 x 10-5 284 2.94 900 Fe 1100 2.4 x 10-12 1.7 x 10-10 6.2 x 10-7 a-Fe 80 0.83 500 900 5.9 x 10-12 5.3 x 10-11 2.3 x 10-3 r-Fe 148 1.53 900 1100 4.2 x 10-19 Cu Cu 7.8 x 10-5 211 2.19 500 18 2.4 x 10-5 4.0 x 10- Zn Cu 189 1.96 500 2.3 x 10-4 6.5 x 10-5 Al 144 500 Al 1.49 4.2 x 10-14 4.1 x 10-14 Cu Al 136 1.41 500 10-4 Mg Al 1.2 131 1.35 500 1.9 x 10-13 2.7 x 10-5 1.3 x 10-22 Cu Ni 256 2.65 500
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 47% (19 reviews)
In order to determine the temperature to which the diffusio...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
A diffusion couple similar to that shown in Figure 5.1a is prepared using two hypothetical metals A and B. After a 30-h heat treatment at 1000 K (and subsequently cooling to room temperature) the...
-
A voltaic cell similar to that shown in Figure 20.5 is constructed. One electrode half-cell consists of a silver strip placed in a solution of AgNO3, and the other has an iron strip placed in a...
-
A voltaic cell similar to that shown in Figure 20.5 is constructed. One half-cell consists of an aluminum strip placed in a solution of Al(NO3)3, and the other has a nickel strip placed in a solution...
-
Write a function my_ieee_2_dec(ieee), where icce is a string contains 64 char- acters of ones and zeros representing a 64-bit IEEE754 number. The output should be d, the equivalent decimal...
-
Bosh Company's assets total $155,000 and its liabilities total $85,000. What is the amount of Bosh's retained earnings if its capital stock amounts to $50,000?
-
On September 28, 2016, a fire destroyed the entire merchandise inventory of Carroll corporation. The following information is available: Sales, January 1-September 28, 2016$560,000 Inventory, January...
-
Refer to your answer to Exercise 11.4 above. Calculate ratios that you believe could be used to interpret the VAS for Buttons Ltd. Explain the purpose of each ratio.
-
Aging of account receivable. Dove company's accounts receivable show the following balance by ages: The credit balance in the Allowance for Uncollectible Accounts is now $17,200. Dove Company's...
-
Futura Company purchases the 72,000 starters that it installs in its standard line of farm tractors from a supplier for the price of $10.40 per unit. Due to a reduction in output, the company now has...
-
Enter a lookup function in cell E5 that returns the tax deduction amount for the number of dependents listed in the cell C5. Use the table in range H13:I17 to complete the function. The maximum...
-
(a) Calculate the diffusion coefficient for copper in aluminum at 500C. (b) What time will be required at 600C to produce the same diffusion result (in terms of concentration at a specific point) as...
-
A diffusion couple similar to that shown in Figure 5.1a is prepared using two hypothetical metals A and B. After a 30-h heat treatment at 1000 K (and subsequently cooling to room temperature) the...
-
A standard signal range for electronic instrumentation is 420 mA. For purposes of monitoring instruments using limit checks, would it be preferable to have an instrument range of 020 mA? Justify your...
-
If bond interests rates fluctuate, but an investor buys and holds their bond until maturity, will the return be equal to, less than, or more than the market rate of interest at the time the bond...
-
What semi-annual payment at the end of each 6 month period is required to pay off a loan of $8000 in ten years if interest is 10% compounded semi-annually?
-
Estimate the initial elastic modulus E, at a temperature of 30C, of an elastomer having a molecular weight of 12,000 gm/mol between crosslinks and a density of 1.0 g/cm 3 .
-
You finance a development with an interest only mortgage having an initial balance of $800 000 to construct a warehouse. The market for warehouse space will either boom or bust next month each...
-
Beta Construction Ltd . acquired the following property, plant and equipment on January 1 , 2 0 1 5 :ResidualAssetOffice equipmentBuildingDelivery equipmentCost$ 7 5 . 0 0 0 2 0 0 , 0 0 0 2 5 0 , 0 0...
-
In Example 9.17, the alternate hypothesis was H1: 3.5 and the P-value for the two-tailed test was P = 0.0256. What would the P-value be for the alternate hypothesis H1: Example 9.17 Perform a...
-
Wal-Mart is the second largest retailer in the world. The data file on the disk holds monthly data on Wal-Marts revenue, along with several possibly related economic variables. a) Using computer...
-
Find the currents flowing in the circuit in Figure 21.50. Explicitly show how you follow the steps in the Problem-Solving Strategies for Series and Parallel Resistors. R 5.0 a R 78 b 14 T; = 24.0 V...
-
Using the Molecule Definition Utility found in both "Metallic Crystal Structures and Crystallography" and "Ceramic Crystal Structures" modules of VMSE, located on the book's web site...
-
Calculate the density of FeO, given that it has the rock salt crystal structure.
-
Magnesium oxide has the rock salt crystal structure and a density of 3.58 g/cm3. (a) Determine the unit cell edge length. (b) How does this result compare with the edge length as determined from the...
-
Prior period adjustments are corrections of material errors in past financial statements. True or False True False
-
Sc. Assume that the standard hours allowed for the actual total output of the fabric plant are 115,000. Calculate the following variances (round to whole dollar): Enter all amounts as positive...
-
Prepare journal entries to record the following transactions relating to long-term bonds of Grier, Inc. (Show computations.) (a) On June 1, 2006. Grier, Inc. issued $600,000, 6% bonds for $587.640,...

Study smarter with the SolutionInn App


