A tert-l3utyI esters [RCO2C (CH3)3] are converted into carboxylic acids (RCO2H) by reaction with trifluoroacetic acid, a
Question:
A tert-l3utyI esters [RCO2C (CH3)3] are converted into carboxylic acids (RCO2H) by reaction with trifluoroacetic acid, a reaction useful in protein synthesis (Section 26.7). Assign E, Z designation to the double bonds of both reactant and product in the following scheme, and explain why there is an apparent change of double-bonds tereochemistry:
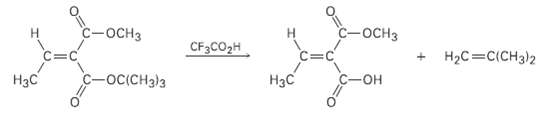
Transcribed Image Text:
с-оснз С-оснз НаС%3DCICH3)2 CF3CO2H Нас С-Он Нас -oc(CH3)3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
Treatment of the tertbutyl ester with trifluoroacetic acid cleaves the OCCH33 group and ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Wonder Textile has 10-year-old sewing machine with a cost price of $5,000. The rate of depreciation on the sewing machine is 10% per annum. The scrap value is $750. It has not been used in the...
-
Explain why firms exist. Explain why there is not just one huge firm.
-
Explain why there is a different money multiplier for each definition of the money supply.
-
A 110 g hockey puck sent sliding over ice is stopped in 15 m by the frictional force on it from the ice. (a) If its initial speed is 6.0m/s, what is the magnitude of the frictional force? (b) What is...
-
Suppose a college student graduates with a major for which he or she lacks enthusiasm. What might this person do about becoming a passionate leader?
-
Summarize the key provisions of NAFTA.
-
Follow the principal steps in preparing a master budget.
-
Refer to Apples financial statements in Appendix A to answer the following. 1. Using fiscal 2011 as the base year, compute trend percents for fiscal years 2011, 2012, and 2013 for net sales, cost of...
-
View Policies Current Attempt in Progress These expenditures were incurred by Bramble Company in purchasing land; cash price $83,000, assumed accrued taxes $2,500, attorneys' fees $3,600, real estate...
-
One of the makeup kit models that Tiegs Corporation produces is targeted at professional women. The goal for Year 2 is to reduce direct materials usage per unit. No defective units are currently...
-
Which of the following E, Z designations are correct, and which are in correct? (a) CH (b) CH2CH=CH2 C=C CH2CH(CH3)2 o C=C (c) Br CH2NH2 C=c CH2NHCH3 (d) NC C C=C (CH3/2NCH2 CH-CH (0 2H c=c (e)...
-
Each of the following carbocations can rearrange to a more stable ion. Propose structures for the likely rearrangement products. H, (a) CH3CH2CH2CH2* (b) CH3CHCHCH3 CH CH CH2* (c)
-
An analysis of the income statement and the balance sheet accounts of Hampton, Inc., at December 31, 2015, provides the following information: Income statement items: Gain on Sale of Marketable...
-
Stealth bombers cruise at an altitude of 1 5 , 0 0 0 meters ( 5 0 , 0 0 0 feet ) where the air temperature is - 6 0 . 0 degrees C . What is the \"speed limit \" of a stealth bomber if we assume they...
-
New Morning Bakery is in the process of closing its operations. It sold its two-year-old bakery ovens to Great Harvest Bakery for $600,000. The ovens originally cost $800,000, had an estimated...
-
The King Carpet Company has $3,030,000 in cash and a total of $11,960,000 in current assets. The firm's current liabilities equal $6,920,000 such that the firm's current ratio equals 1.7. The...
-
Sunland Co. uses a periodic inventory system. Its records show the following for the month of May, in which 73 units were sold. Units Unit Cost Total Cost May 1 Inventory 34 $12 $408 15 Purchases 27...
-
Charies is 65 years of age and in receipt of a CCP retirement pension, He files a CPT30 election form on April 15th to stop CPP contributions: his employer stopped deducting CPP contributions as of...
-
Trinity Church has been asked to operate a homeless shelter in its parish hall. To operate a homeless shelter, the church would have to hire a full-time employee for\($2,200\) per month to manage the...
-
Identify the most stable compound:
-
How do the properties of water differ from those of most other substances?
-
What alkenes would give each of the following alcohols as the major (or only) product as a result of oxymercuration-reduction? CH-CH
-
Give the product(s) expected from the hydroboration--oxidation of each of the following alkenes. (a) Cyclohexene lel trans-4-methyl-2-pentene (b) Cis-3-hexene
-
Contrast the answers for Problem 5.8 with the answers for the corresponding parts of Problem 5.5. For which alkenes are the alcohol products the same? For which are they different? Explain why the...
-
If funds are withdrawn from an Oregon 529 College Savings Network plan for qualified education purposes, then which of the following is correct? Question 8Select one: a. The taxpayer will have an...
-
Nancy is single, 19 years old and a full-time college student in Oregon. Her parents claim her as a dependent. Nancy had earned and gross income of $820.00 for the year. Will Nancy be required to...
-
Use the australian accounting standard. On 1 July 2022, Surf Ltd leases a semi-trailer truck from Superior Ltd. The truck will be used to move spare parts between Surf's various warehouses. The...

Study smarter with the SolutionInn App


