Addition of HBr to 1-phenyipropene yields only (1-bromopropyl) benzene. Propose a mechanism for the reaction, and explain
Question:
Addition of HBr to 1-phenyipropene yields only (1-bromopropyl) benzene. Propose a mechanism for the reaction, and explain why none of the other regioisomer isproduced.
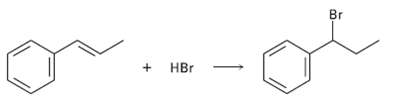
Transcribed Image Text:
Br + HBr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
HBr o Protonation of the double bond ...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the reaction of acetyl chloride with phenylmagnesium bromide to give 1,1-diphenylethanol. OH (1) ether solvent CH3-C CI 2 (2) H 0 acetyl chloride phenylmagnesium bromide...
-
Propose a mechanism for the reaction of cyclohexyl methyl ketone with excess bromine in the presence of sodium hydroxide.
-
Propose a mechanism for the reaction of an amide with thionyl chloride to form a nitrile.
-
How did your parents communication with you influence your self-concept?
-
Sam, age 45, is single. For 2016, he has the following items: Business income........................................................$70,000 Business...
-
Why do companies like to issue convertible securities? Whats in it for them?
-
Using the data in questions 12 and 13, what is the overhead volume variance for June? Is the variance favorable or unfavorable?
-
Tarheel Furniture Company is planning to establish a wholly owned subsidiary to manufacture upholstery fabrics. Tarheel expects to earn $1 million after taxes on the venture during the first year....
-
Based on the following data for the current year, what is the number of days' sales in inventory? Net sales on account during year $484,710 Cost of goods sold during year 153,008 Accounts receivable,...
-
Each of the following independent events requires a year-end adjusting entry. Record each event and the related adjusting entry in general journal format. The first event is recorded as an example....
-
Electrophilic substitution on 3-phenylpropanenitrile occurs at the ortho and Para positions, but reaction with 3-phenylpropenenitrile occurs at the meta position. Explain using resonance structures...
-
Triphenylmethane can be prepared by reaction of benzene and chloroform in the presence of A1Cl3. Propose a mechanism for thereaction. H. + CHCI3 AICI3
-
An insulated rigid tank is divided into two equal parts by a partition. Initially, one part contains 4 kg of an ideal gas at 800 kPa and 50oC, and the other part is evacuated. The partition is now...
-
Draw an inelastic supply curve, give an example of a good with a relatively inelastic supply? Draw an elastic supply curve, give an example of a good with a relatively elastic supply? Draw an...
-
10. The isotope deuterium has a. one proton. b. one proton and one neutron. c. one proton and two neutrons. d. one proton and three neutrons. 11. After filling the first shell, the outermost shell of...
-
A data set has the regression equation y = -3.5x+1500 What is the residual value for the data value of (450, 50)?
-
Develop and explain potential solutions for a laboratory improvement project. Where nonconformance has been steadily rising from 2017 to September 2019 Year Type of Nonconformance Laboratory...
-
Matthew Young Company sells 9% bonds having a maturity value of $2,360,000 for $2,104,790. The bonds are dated January 1, 2025, and mature January 1, 2030. Interest is payable annually on January 1....
-
White a briefmemo that distinguishes between the dements of financial statements and the accounts that appear on financial statements.
-
(a) Given a mean free path = 0.4 nm and a mean speed vav = 1.17 105 m/s for the current flow in copper at a temperature of 300 K, calculate the classical value for the resistivity of copper. (b)...
-
Derive the following Maxwell relations for open systems. a. Starting from Eq. 6.2-5a, b. Starting from Eq. 6.2-6a, c. Starting from Eq. 6.2-7a, d. Starting from Eq. 6.2-8a, (). HT ON (0) aN S, N S,V...
-
Show the products of thesereactions: CH NaOH Br2 Bra b) a) . .
-
The reaction of an alkenes with bromine in an alcohol as solvent produces as ether as the product. Show a mechanism for the following reaction and explain the stereochemistry of theproduct. Br . H....
-
Show all the steps in the mechanism for the formation of MTBE from methanol and isobutylene.
-
Jon est lidiando con varios empleados que siempre llegan tarde. Decide unilateralmente descontar el pago diario de todos en un 5 por ciento si llegan ms de 10 minutos tarde. Jon tendr mejores...
-
People sometimes apply the Cost-Benefit Principle incorrectly because they evaluate costs and benefits ______. Multiple choice question. as proportions rather than absolute dollar amounts as absolute...
-
Bob, an employee of UNC, is contemplating the amount to contribute to the flexible medical spending account for the next calendar year. Based on past medical expenses, and projections of medical...

Study smarter with the SolutionInn App


