Aldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield
Question:
Aldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:
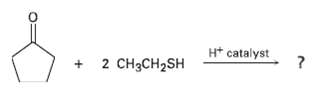
Transcribed Image Text:
H* catalyst 2 CH3CH2SH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
The same series of steps used to form an acetal is followed in thi...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the product of the following reaction. M 1) LIAID, 2) H20 Me
-
Predict the product of the following reaction. Br NaOCH3, heat -NO2
-
Predict the product of the reaction if CH3-S-CH2CH2CH2CH2-Br is heated in a polar organic solvent, such as methanol. Similarly, what would be the product for hexyl bromide in methanol? (In hexyl...
-
1. Review the six (6) goals of performance based acquisition(s); choose two and discuss how your choices can be implemented for effective management of contracts, using an actual or theoretical...
-
1. What factors of group cohesiveness were present in this team? 2. What should Joe do now to ensure the team's effectiveness and ultimate success? Use the Model of Team Effectiveness (PPT 15-9) to...
-
Rationalize the following lattice energy values: Lattice Energy (k/mol) -2862 -2130 -2721 -2095 Compound CaSe Na2Se CaTe NazTe
-
How does the existence of classic and integrated corporate tax systems affect the financing decisions of multinational corporations?
-
On January 1, 2020, Prasad SpA had the following equity accounts. Share CapitalOrdinary (25 par value, 48,000 shares issued and outstanding) .......................1,200,000 Share PremiumOrdinary...
-
Mc 2 1 0103.56 eBook Matthew Flaws is a single taxpayer and lives at 5670 Sierra Drive, Honolulu, HI 96822. He has income from a job as a marketing representative, Interest income, dividend income,...
-
Leighton Industries needed steel pipe to build furnaces for a customer. Leighton sent Callier Steel an order for a certain quantity of A 106 Grade B steel. Callier confirmed the order and created a...
-
6-Methyl-5-hepten-2-one is a constituent of lemongrass oil. How could you synthesize this substance from methyl4-oxopentanoate? CH3CH2CH2OCH3 Methyl 4-oxopentanoate
-
Ketones react with dimethyl-sulfonium methylide to yield epoxides. Suggest a mechanism (or the reaction. HICHJ)2 DMSO solvent (CH3)2S Dimethylsulfonium methylide
-
Consider Problem 10, Set 2.3e (Chapter 2). (a) Which of the specification constraints impacts the optimum solution adversely? (b) What is the most the company should pay per ton of each ore?
-
Briefly explain the services provided by the data link layer. ( ii ) Identify the two sub - layers of the data link layer and state function of each in data transmission. ( iii ) Given the MAC...
-
Cranbrook Used Books experienced the following events during the current year: a. Incurred $12,000 in selling costs. b. Took an inventory at year-end and learned that goods costing $100,000 were on...
-
15 Following are some transactions and events of Business Solutions. February 26 The company paid cash to Lyn Addie for eight days' work at $120 per day. March 25 The company sold merchandise with a...
-
Question: You are lucky enough to be involved with putting together the Olympic bid for the Summer Olympic Games in 2032. You have been asked to help assemble the bid documents for six of the...
-
4. In the Bag-of-features algorithm (Video Google) two feature detectors were used: Shape Adapted and Maximally Stable Extremal Regions. (a) What was the reason for this choice and what is each one...
-
The number of births per woman in China is 1.6 down from 5.91 in 1966. This fertility rate has been attributed to the law passed in 1979 restricting births to one per woman. Suppose that a group of...
-
Phosgene, COCl2, is a toxic gas used in the manufacture of urethane plastics. The gas dissociates at high temperature. At 400oC, the equilibrium constant Kc is 8.05 104. Find the percentage of...
-
Explain why it is not necessary to find the Lewis structure with the smallest formal charges to make a successful prediction of molecular geometry in the VSEPR theory. For example, write Lewis...
-
Write the mechanistic steps in the cyclization of ethyl phenylacetoacetate (ethyl 3-oxo-4-phenylbutanoate) in concentrated sulfuric acid to form naphthoresorcinol (1,3-naphthalenediol).
-
When an aldehyde or a ketone is condensed with ethyl a-chloroacetate in the presence of sodium ethoxide, the product is an α,β-epoxy ester called a glycidic ester. The...
-
The Perkin condensation is an aldol-type condensation in which an aromatic aldehyde (ArCHO) reacts with a carboxylic acid anhydride, (RCH2CO)2O, to give an a,b-unsaturated acid (ArCH "CRCO2H). The...
-
The CRA requires an employer to submit: Multiple Choice A T4 summary for the employer for the reporting year A T4 information slip for each employee with earnings in the reporting year and A T4...
-
The cash ratio is computed by dividing the sum of cash and cash equivalents by Blank______
-
Accounting I9) Wood and Lamb Furniture company had net Accounts Receivable of $790,000 at thebeginning of the year and $945,000 at the end of the year. Net Sales Revenue for 2010was $6,540,000. What...

Study smarter with the SolutionInn App


