Aldehydes can be prepared by the Wittig reaction using (methoxymethylene)-triphenylphosphorane as the Wittig reagent and then hydrolyzing
Question:
Aldehydes can be prepared by the Wittig reaction using (methoxymethylene)-triphenylphosphorane as the Wittig reagent and then hydrolyzing the product with acid. For example,
(a) How would you prepare the necessary phosphorane?
(b) Propose a mechanism for the hydrolysisstep.
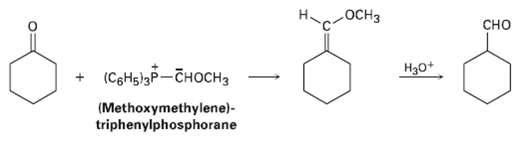
Transcribed Image Text:
OCH3 сно (СоНiзр— сноснз H30+ (Methoxymethylene)- triphenylphosphorane
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
a b Ph3P BrCHOCH3 HOCH3 H0H protonation ...View the full answer

Answered By

Sinmon Warui Kamau
After moving up and down looking for a job, a friend introduced me to freelance writing. I started with content writing and later navigated to academic writing. I love writing because apart from making a living out of it, it is also a method of learning and helping others to learn.
5.00+
40+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you prepare 72.5 g of an aqueous solution that is 5.00% potassium iodide, KI, by mass?
-
How would you prepare 455 g of an aqueous solution that is 6.50% sodium sulfate, Na2SO4, by mass?
-
How would you prepare 2.00 L of (a) 0.10 M KOH from the solid? (b) 0.010 M Ba(OH)2 8H2O from the solid? (c) 0.150 M HCl from a reagent that has a density of 1.0579 g/mL and is 11.50% HCl (w/w)?
-
Every human being fears to fail. This is the same case with the leaders. Most of the leaders are afraid of some things, but failing is the most feared among all of them. The loss of direction among...
-
Various conflict management styles are depicted on PPT 9-7. For each of the participants in the scenario, describe which style(s) they used and cite examples to support your answer.
-
A certain microwave oven delivers 750 watts (J/s) of power to a coffee cup containing 50.0 g of water at 25.0oC. If the wavelength of microwaves in the oven is 9.75 cm, how long does it take, and how...
-
As a U. S. congressman from South Carolina you are considering proposing a bill to eliminate a state income tax of 7 percent and a sales tax of 5 percent and replace it with a value-added tax of 10...
-
Shank Company manufactures candy. On September 1, Shank purchased a futures contract that obligates it to sell 150,000 pounds of sugar on September 30 at $0.41 per pound. Shank typically purchases...
-
Equipment which cost $437000 and had accumulated depreciation of $236000 was sold for $226000. This transaction should be shown on the statement of cash flows (indirect method) as a(n) addition to...
-
You have an obligation to pay $1,000,000 in 3 years from now, and you would like to make an investment now that will enable you to meet this obligation. This investment will be a portfolio containing...
-
How would you use a Grignard reaction on an aldehyde or ketone to synthesize the following compounds? (a) 2-Pentanol (b) 1-Butanol (c) 1-Phenylcyclohexanol (d) Diphenyl methanol
-
When 4-hydroxyhutanal is treated with methanol in the presence of an acid catalyst, 2-mcthoxytctrahydrofuran is formed. Explain. -H C HCH2H2CH C
-
For the following exercises, use the given information to find the equation for the ellipse. A whispering gallery is to be constructed such that the foci are located 35 feet from the center. If the...
-
Swifty Enterprises sold $740,000 of accounts receivable to Wildhorse Factors, Inc. on a without recourse basis under IFRS, as the risks and rewards have been transferred to Wildhorse. The transaction...
-
Tax Evaluation - Beth Johnson's Income from Partnership This activity provides hands - on experience in assessing and reporting partnership incomes, offering insights into the intricacies of tax...
-
How does the intricate interplay between technological obsolescence, innovation diffusion, and disruptive paradigms delineate the contours of the contemporary technology cycle, and what implications...
-
The operations vice president of Home Bank has been interested in investigating the efficiency of the bank's operations. She has been particularly concerned about the costs of handling routine...
-
Graph the function. 31 for x
-
Suppose an airline claims that its flights are consistently on time with an average delay of at most 15 minutes. It claims that the average delay is so consistent that the variance is no more than...
-
1-Stern observed all of the following results EXCEPT _______ in his experiment. A-one of the recombinant phenotypes was associated with an X chromosome of normal length B-the number of car, B+ male...
-
The greatest bond length is found in (a) O 2 ; (b) N 2 ; (c) Br 2 ; (d) BrCl.
-
When acrolein (propenal) reacts with hydrazine, the product is a dihydropyrazole: Suggest a mechanism that explains this reaction. H + H2N-NH2 Acrolein Hydrazine A dihydropyrazole
-
(a) Propose step-by-step mechanisms for both transformations of the Robinson annulations sequence just shown. (b) Would you expect 2-methylcyclohexane-1, 3-dione to be more or less acidic than...
-
Outline reasonable mechanisms that account for the products of the following Mannich reactions: (a) (b) (c) NMe2 Me2N NMe2 + 2 Me2NH CH3 CH
-
In the dividends-based valuation process discussed in the book and my notes, what does "continuing value" refer to? The value of dividends after the forecast horizon, which in book examples is...
-
The right side of the manufacturing overhead account Blank______. Multiple select question. will usually equal the left side of the account at the end of the year accumulates overhead costs as they...
-
when a practice recognizes and records revenue only when payments or cash are received by the practice and only recognizes and reports an expense when money is actually paid out, they are using what...

Study smarter with the SolutionInn App


