All naturally occurring amino acids have the same relative configuration. All have the S absolute configuration, except
Question:
All naturally occurring amino acids have the same relative configuration. All have the S absolute configuration, except for cysteine, which has the R configuration.Explain.
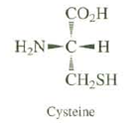
Transcribed Image Text:
CO.H H,N-C-H CH,SH Cysteine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (17 reviews)
In all of the amino acids except cysteine the CO 2 H group has priority ...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Most naturally occurring amino acids have chirality centers (the asymmetric carbon atoms) that are named (S) by the Cahn-Ingold-Prelog convention (Section 5-3). The common naturally occurring form of...
-
The 20 naturally occurring amino acids (See the following table) are all l amino acids, and they all have the S configuration, with the exception of glycine (which lacks a chirality center) and...
-
Identify which 2 of the 20 naturally occurring amino acids are expected to have the same pI.
-
1. As a policy maker you should never worry much about those are eligible for Medicaid benefits and do not enroll. This is because they will enroll in public insurance if they need it. True or False?...
-
Classify the following organisations as professional service firms, mass service entities or service shops. In each case, explain the reasons for your classification. 1. Motor registration department...
-
You are considering an investment in Roxies Bed & Breakfast Corp. During the last year the firms income statement listed an addition to retained earnings of $4.8 million and common stock dividends of...
-
A \(20 \%\) discount is offered on a new laptop. How much is the discount if the new laptop originally cost \(\$ 700\) ?
-
Answer each of the questions in the following unrelated situations. (a) The current ratio of a company is 5:1 and its acid-test ratio is 1:1. If the inventories and prepaid items amount to $500,000,...
-
Please answer the following question. Thank you very much for your assistance and hope you have a good evening. Thanks again.. SV 1 point On January 2, 2021, Ross Co. purchased a machine for $500....
-
Majka Company was started on January 1, Year 1. During Year 1, the company experienced the following three accounting events: (1) earned cash revenues of $28,600, (2) paid cash expenses of $13,200,...
-
Draw a stereo isomer of this compound that is chiral, and draw two that are not chiral? CH3 -
-
Explain whether each of these compounds is chiral or not: , CCH3 C=C=C] CH3 . a) b) C=C=C, , , . c) CH;O. `CH3 - . d) CH;O CO,H CH;OS Br CH;CH2 e) f) N-CH,CH3 Ph CH;CH2
-
Unilever Group reports the following equity information for the years ended December 31, 2009 and 2010 (euros in millions). 1. For each of the three account titles share capital, share premium, and...
-
(a) Explain the law of variable proportions and its usefulness to the producer. Illustrate with a relevant diagram (7marks) (b) Explain the features of an oligopoly market perfectly competitive...
-
An insulated beaker with negligible mass contains liquid water with a mass of 0.240 kg and a temperature of 65.1 C. 9 Part A How much ice at a temperature of -10.8 C must be dropped into the water so...
-
You are asked to design an automotive radiator using a 10 mm diameter circular tube. You need to know heat loss per unit length of the cylinder. Other conditions of design are: Air pressure of 101...
-
Find the Charge on the Spheres Two identical small charged spheres, each having a mass of 7.0 x 10-2 kg, hang in equilibrium as shown in figure (a). The length L of each string is 0.100 m, and the...
-
Pawsitive Medical Supplies Sdn. Bhd. (PMSSB) was incorporated in the year 2017 with a factory in Ayer Hitam Industrial Park, Johor. It manufactures veterinary medical devices, which are listed by the...
-
Three inverting amplifiers, each with \(R_{2}=150 \mathrm{k} \Omega\) and \(R_{1}=15 \mathrm{k} \Omega\), are connected in cascade. Each op-amp has a low-frequency gain of \(A_{o}=5 \times 10^{4}\)...
-
Suppose Green Network Energy needs to raise money to finance its new manufacturing facility, but their CFO does not think the company is financially capable of making the periodic interest payments...
-
Simplify each expression. 7+2(-10x + 4)
-
Ribose, an essential part of ribonucleic acid (RNA), has the following structure: (a) How many chirality centers does ribose have? Identify them. (b) How many stereo isomers of ribose are there? (c)...
-
On catalytic hydrogenation over a platinum catalyst, ribose (Problem 9.57) is converted into ribitol. Is ribitol optically active or inactive?Explain. CH2 Ribitol
-
Hydroxylation of cis-2-hutene with OsO4 yields 2, 3-butanediol. What stereochemistry do you expect for the product?
-
help please The following information pertains to Carla Vista Company. 1. Cash balance per books, August 31, \( \$ 7,424 \). 2. Cash balance per bank, August 31, \( \$ 7,388 \). 3. Outstanding...
-
Deming & Sons manufactures four grades of lubricant, W-10, W-20, W-30, and W-40, from a joint process. Additional information follows: Units Sales Value at Product Produced Split-Off W-10 63,000 $...
-
Chapter 10 Performance Evaluation Using Variances from Standard Costs 471 c. Controllable variance, RM180 U PR 10-3 Direct materials, direct labor, and factory overhead cost variance analysis OBJ. 3,...

Study smarter with the SolutionInn App


