Amino acids such as alanine actually exist as species called zwitterions, with a positive charge on the
Question:
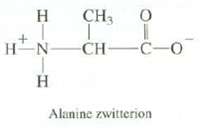
Amino acids such as alanine actually exist as species called zwitterions, with a positive charge on the nitrogen and a negative charge on the oxygen. Explain what effect you expect this to have on the melting point of alanine.
Transcribed Image Text:
CH3 0 T HN CH- HINIH Н Н I -C-0 Alanine zwitterion
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (17 reviews)
The zwitterions form of al...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Why are amino acids such as arginine and histidine required in relatively large amounts by children but in smaller amounts by adults? The adult human is not able to make these amino acids.
-
Calculate the formal charge on the nitrogen atom in ammonia, NH3; in the ammonium ion, NH4+; and in the amide ion, NH2.
-
If you painted positive charge on the floor, what surface charge density would be necessary to suspend a 15C, 5.0-g particle above the floor?
-
Consider a soap bubble. Is the pressure inside the bubble higher or lower than the pressure outside?
-
The 'Peal life' suggests that senior executive salaries have risen considerably over time. What do Herzberg's theory of motivation and expectancy theory suggest about using large salary packages to...
-
Plato Energy is an oil and gas exploration and development company located in Farmington, New Mexico. The company drills shallow wells in hopes of finding significant oil and gas deposits. The firm...
-
Spratt Co. (a U.S. firm) attempts to determine its economic exposure to movements in the British pound by applying regression analysis to data over the last 36 quarters: where SP represents the...
-
What is meant by the term transferred-out cost? Why does the transferred-out cost under the WA method include only one computation but the FIFO method includes multiple computations?
-
4 Listen (2) = 3z-15 25-2 has one vertical asymptote, on The graph of the function H hole, and a horizontal asymptote. The vertical asymptote equation is = A (Type an integer or a simplified...
-
a. Keith Thomas and Thomas Brooks began a new consulting business on January 1, 2016. They organized the business as a C corporation, KT, Inc. During 2016, the corporation was successful and...
-
Glucose is a typical carbohydrate. What functional groups are present in glucose? What would you predict about the water solubility of glucose, oiler a reason why nature uses carbohydrates rather...
-
What is the DU of estrone, a female sex hormone? Use the number of carbons and oxygen?s in estrone to calculate the number of hydrogen?s it has. Estrone 0 CH3
-
The earning of revenue by a business enterprise is recognized for accounting purposes when the transaction is recorded. In some situations, revenue is recognized approximately as it is earned in the...
-
3. What are the potential pitfalls for the underwriter of all the information made available by technology
-
(30) Nelson Corporation sells three different products. The following information is available on December 31: Inventory Item Units X Y Z Cost per unit Net realizable value per unit 300 $4.00 $3.00...
-
what is the fair price of te futures contract based on the s&p500 which expires in 3 months time
-
You're feeling particularly lazy and decide to open a door by firing a bullet at it. The door has a mass of 14.5 kg and is 0.950 m wide, and the 10 g bullet, traveling at 470. m/s, hits the door at...
-
John Lemon, a fund manager currently holds $100 million par value of Treasury note T-A. He is worried that the interest rate might increase in the near future thus thinking of hedging his position...
-
If an interest rate of 3.72 per cent is quoted on a 360-day count convention, what is the bey rate for this?
-
2.) Find the Laplace transform of f(t) 7e-St cos 2t +9 sinh2 2t. Use Laplace Table. %3D
-
Show that the average velocity of a car over a time interval [t 1 , t 2 ] is the same as the average of its velocities during the trip.
-
TMS ethers can be removed by treatment with fluoride ion as well as be acid-catalyzed hydrolysis. Propose a mechanism for the reaction of cyclohexyl TMS ether with LiF. Fluorotrimethyisilane is a...
-
Show the mechanism of the reaction of p-methaylphenol with 2-methylpropene and H3PO4 catalyst to yield the food additive BHT.
-
Assume that you need to prepare 5-cholesten-3-one from cholesterol. How could you use IR spectroscopy to tell whether the reaction was successful? What differences would you look for in the IR...
-
The adjusted trial balance for Tybalt Construction on December 31 of the current year follows. TYBALT CONSTRUCTION Adjusted Trial Balance December 31 Number 101 Account Title Debit Credit Cash $...
-
Wendell's Donut Shoppe is investigating the purchase of a new $39,600 donut-making machine. The new machine would permit the company to reduce the amount of part-time help needed, at a cost savings...
-
D Question 21 In all of the following problems, use these tax rates and taxable wage: OASDI -6.2% on the first $137,7700; HI-1.45% on all taxable wages. Beginning with the first pay of the year,...

Study smarter with the SolutionInn App


