Ammonia, NH3, has pKa = 36 and acetone has pKa 19. Will the following reaction takeplace? Na*
Question:
Ammonia, NH3, has pKa = 36 and acetone has pKa 19. Will the following reaction takeplace?
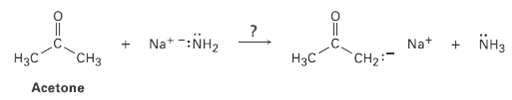
Transcribed Image Text:
Na* -:NH2 NH3 CHз Нас Hзс Acetone Na+ CH2:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
CH3 NaNH HC pK 19 Stro...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Under standard conditions, will the following reaction proceed spontaneously as written? Fumarate NADHHsuccinate NAD 1
-
Under standard conditions, will the following reaction proceed spontaneously as written? Cyto a(Fe + cyto b(Fe) oto a(Fe")+ ono b(Fe) 3 +
-
Ammonia and hydrogen chloride react to form solid ammonium chloride: NH3 (g) + HCl (g) NH4CL (s) Two 2.00-L flasks at 25oC are connected by a valve, as shown in the drawing on the next page. One...
-
Find a dense document that has little white space and few headings or graphics. You might look at a legal court filing, a companys privacy statement, or an apartment lease. How could you redesign the...
-
On August 31, 2015, Harvey and Margaret, who file a joint return and live in Charleston, South Carolina, sell their personal residence, which they have owned and lived in for 10 years. The realized...
-
The ACT is a college entrance exam. In addition to administering this exam, researchers at ACT gauge high school students readiness for college-level subjects. For example, ACT has determined that a...
-
Caan Oil Company is considering investing in a new oil well. It is expected that the oil well will increase annual revenues by \($120,000\) and will increase annual expenses by \($80,000\) including...
-
Marias Food Service provides meals that nonprofit organizations distribute to handicapped and elderly people. Here is her forecasted income statement for April, when she expects to produce and sell...
-
Morgan Taylor is settling a $18,000 loan due today by making 6 equal annual payments of $3,893.68. Click here to view factor tables Determine the interest rate on this loan, if the payments begin one...
-
Atlantic Life Ltd. is in the business of selling a life insurance company. The corporation hired Toby Ryan as its representative in Newfoundland. Toby was provided with Atlantic Life business cards...
-
Will either of the following reactions take place as written, according to the data inTable? (a) HCN + CH3CO2 Na+ Na* "CN CHCO2H Na* "CN CH3CH20- Na* |(b) CH3CH2OH HCN
-
What is the Ka of HCN is its pKa = 9.31?
-
Nitric acid hydrates have received much attention as possible catalysts for heterogeneous reactions that bring about the Antarctic ozone hole. Worsnop et al. investigated the thermodynamic stability...
-
A 48 kg space probe orbits a newly discovered planet. Its orbital radius is 5.46x10 6 m and the mass of the planet is 2.23x10 24 kg. The space probe contains two devices- a mass attached to a spring...
-
Read the case information and writea2-3page paper in which you: choose any case: https://www.chronline.com/crime/ Summarize the facts of the case. Examine how the procedure would differ if the...
-
Teams are now used to produce goods and services, to manage projects, and to make decisions about running the company. by only a few O primarily by small O primarily by large by almost all O...
-
Two masses, m 1 = 3.29 kg and m 2 = 8.00 kg, are connect by a string of negligible mass. The string passes over a frictionless pulley so that m 1 and m 2 hang down on opposite sides of the pulley....
-
One of the isotopes Madame Curie studied, radium-226, has a half-life of 1600 years. Determine how many years are required for amount of radium-226 on the remains of Madame Curie to reduce to 3% of...
-
On March 6, 2006, Lies Imports purchased merchandise from The Glass Exchange with a list price of \($15,500\), terms 2/10, n/45. On March 10, Lies returned merchandise to The Glass Exchange for...
-
Research corporate acquisitions using Web resources and then answer the following questions: Why do firms purchase other corporations? Do firms pay too much for the acquired corporation? Why do so...
-
a. Derive Eq. 7.4-8. b. Derive Eq. 7.4-12. c. Obtain an expression for the fugacity of a pure species that obeys the van der Waals equation of state in terms of Z, B = P b/RT, and A = aP/(RT) 2...
-
Draw structures for these compounds. (a) 2-Methylcylohexanol (b) 4-Methyl-4-penten-2-ol
-
Name thesecompounds: a) CHOCHCH3 OCH, b) CI
-
Name these compounds: (a) CH 3 CH 2 CH 2 NH 2 b) NHCH,CH,
-
Which of the following is a SOURCE of cash?Group of answer choicesAn increase in Prepaid TaxesThe purchase of a new truckA decrease in Accounts PayableThe sale of marketable securities
-
Dec. 1, 2024: Mortgaged the warehouse for $550,000 cash with Southside Bank. The mortgage requires monthly payments of $7,000. The interest rate onthe note is 3% and accrues monthly. The first...
-
According to the Code, the subscriber must make the following disclosures: Group of answer choices Information about fees and charges. Information about fees and charges and daily or periodic...

Study smarter with the SolutionInn App


