Assign as many of the resonances as you can to specific carbon atoms in the 13C NMR
Question:
Assign as many of the resonances as you can to specific carbon atoms in the 13C NMR spectrum of ethylbenzoate.
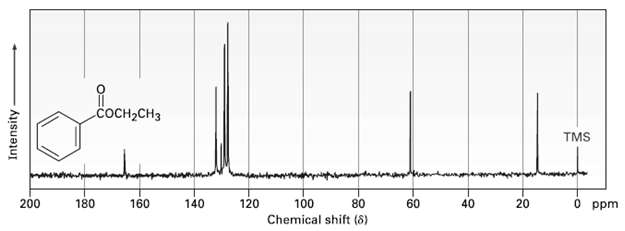
Transcribed Image Text:
соCH-CHз TMS 100 Chemical shift (8) 200 180 140 20 0 ppm 120 40 160 60 Intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (18 reviews)
0 5 10 6 4 5 OO 2 ...View the full answer

Answered By

ZIPPORAH KISIO LUNGI
I have worked on several other sites for more than five years, and I always handle clients work with due diligence and professionalism. Am versed with adequate experience in the fields mentioned above in which have delivered quality papers in research, thesis, essays, blog articles, and so forth.
I have gained extensive experience in assisting students to acquire top grades in biological, business and IT papers. Notwithstanding that, I have 7+ years of experience in corporate world software design and development.
5.00+
194+ Reviews
341+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
3-Methyl-2-butanol has five signals in its 13C NMR spectrum at 17.90, 18.15, 20.00, 35.05, and 72.75 ?. Why are the two methyl groups attached to C3 nonequivalent? Making a molecular model should be...
-
What changes would you expect in the 13C NMR spectrum of 1-bromopropane upon cooling the compound to very low temperature?
-
Assign the resonances in the 13C NMR spectrum of methyl propanoate, CH3CH2CO2CH3(figure). TMS CH-cH 2 1 120 200 180 160 140 100 20 40 O ppm 60 Chemical shift (8) Intensity
-
What is the difference between an optimistic approach and a pessimistic approach to decision making under assumed uncertainty?
-
American and European bricks-and-mortar retailing is increasingly becoming dominated by "hyper marts," enormous stores that sell groceries, household goods, hardware and other products under one...
-
Great Sounds manufactures and sells a new line of radios. Unfortunately, Great Sounds suffered serious fire damage at its home office. As a result, the accounting records for October were partially...
-
A well-known insurance company offers a policy known as the Estate Creator Six Pay. Typically the policy is bought by a parent or grandparent for a child at the childs birth.The details of the policy...
-
Develop an EOQ solution and calculate total relevant costs for the data in the preceding table. Stockout costs equal $10 per unit. *Holding cost = $2.50/unit/week; setup cost = $150; lead time = 1...
-
Due to the relative complexity of quantitative investment models, it is appropriate for CFA members not to discuss the assumptions used in investment models and processes to generate the analysis,...
-
A bicycle wheel of radius R and mass M is at rest against a step of height 3R/4, as illustrated in FIGURE 11-65. Find the minimum horizontal force F that must be applied to the axle to make the wheel...
-
Long-range coupling between protons more than two carbon atoms apart is sometimes observed when ? bonds intervene. An example is found in 1-methoxy-1-buten-3-yne. Not only does the acetylenic proton,...
-
The 1H and 13C NMR spectra of compound A, C8H9Br are shown. Propose a structure for A, and assign peaks in the spectra to your structure. TMS O ppm 10 8. 6. Chemical shift (8) TMS 200 180 160 140 120...
-
When a cylinder rolls on a horizontal surface as in Figure 10.28, do any points on the cylinder have only a vertical component of velocity at some instant? If so, where are they?
-
Discuss the following quotes and ITS concepts back to biblical principles Happiness can be found, even in the darkest of times, if one only remembers to turn on the light. ~ Harry Potter and the...
-
Discuss the types of information which should be included in an officer's field notes taken at the crime scene. Explain the official police report's primary purpose and how the three-pronged rule of...
-
Proverbs 24:10 says, "If you are slack in the day of distress, your strength is limited." What do you do when you are faced with adversity?
-
Why do commercial invoices and packing slips not contain the same information? Explain.
-
Two blocks (mA = 4.3 kg and mg = 5.1 kg) are connected to each other and to a central post by two thin ropes as shown. The blocks rotate on a frictionless horizontal surface in circles of radii (rA =...
-
OPERATIONS WITH FRACTIONS a) b) 415 + 715 23 54 59 e) c) d) 56 47 12 34 32 + 23 88 56 f) 57 ++ g) h) 83 +
-
Suppose that fraction used = / 1.0 + 0.1Mt. for some parameter 1. Write the discrete-time dynamical system and solve for the equilibrium. Sketch a graph of the equilibrium as a function of ....
-
A KNO 3 solution containing 45 g of KNO 3 per 100.0 g of water is cooled from 40 C to 0 C. What happens during cooling?
-
Predict the products of the following reactions. (a) allyl bromide + cyclohexyl magnesium bromide (b) cyclopentadiene + anhydrous HCI (c) 2-methylpropene + NBS, light (d) Furan + trans 1,...
-
Show how the reaction of an allylic halide with a Grignard reagent might be used to synthesize the following hydrocarbons. (a) 5-methylhex-1-ene (b) 2,5,5-trimethylhept-2-ene (c)...
-
Draw the important resonance contributors for the following cations, anions, and radicals. (a) (b) (c) (d) (e) (f) (g) (h) CH CH 2 - CH2 OF OCH3
-
Find a sum-of-products expression for F' for the function F(G, H, I, J) = H+IJ(G+H') Edit View Insert Format Tools Table 12pt v Paragraph B T U AV
-
Glenn and Maria are with two children. Glenn earns $4,500 per year while Maria earns $40,000. They spend $5,000 for dependent care for their children. How much will their dependent care tax credit...
-
Use this graph of the derivative of f. y 5+ 4+ 3+ 2+ 1+ +3 2 +2 ++ -5-4-3-2-1 -1 -2+ -3- -4+ -5+ 1 4 5 x

Study smarter with the SolutionInn App


