Assign R or S configuration to the following molecule, write the product you would expect from SN2
Question:
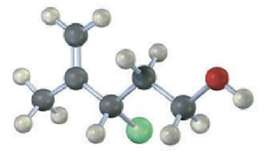
Assign R or S configuration to the following molecule, write the product you would expect from SN2 reaction with NaCN, and assign R or S configuration to the product (yellow-green =Cl):
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
CH HH CHOH NaCN H3C CH HH R NC H CH2OH NaCl HCI T...View the full answer

Answered By

Ashish Bhalla
I have 12 years work experience as Professor for Accounting, Finance and Business related subjects also working as Online Tutor from last 8 years with highly decentralized organizations. I had obtained a B.Com, M.Com, MBA (Finance & Marketing). My research interest areas are Banking Problem & Investment Management. I am highly articulate and effective communicator with excellent team-building and interpersonal skills; work well with individuals at all levels.
4.80+
17+ Reviews
46+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Assign R or S configuration to the chirality center in the following molecular model of the amino acid methionine (blue = N, yellow =S):
-
Assign R or S configuration to the chirality center in each of the followingmolecules: CH (c) . (b) (a) 2 "- HS efpo -- CH-
-
Assign R or S configuration to each chirality center in the followingmolecules: (a) (b) ,CH (c) , . CH CHCH
-
In the Walton Bookstore example with a discrete demand distribution, explain why an order quantity other than one of the possible demands cannot maximize the expected profit.
-
Evaluate Brian Conrad's approach to appraisal interviews. Write a paragraph or two summarizing what Conrad is doing well and how he might further improve the effort.
-
"If the translation of an integrated foreign operation produced a gain, the translation of the same company could produce a loss if the operation were instead considered to be self-sustaining." Do...
-
What is the probability that the dealer is dealt an initial hand worth 21 points, with an ace showing?
-
In an L-R-C series circuit, R = 300, L = 0.400 H, and C = 6.00 X 10-8 F. When the ac source operates at the resonance frequency of the circuit, the current amplitude is 0.500 A. (a) What is the...
-
4. a) At a price of $8 per ticket, a musical theatre group can fill every seat in the theatre, which has a capacity of 1500. For every additional dollar charged, the number of people buying the...
-
with the below question There is no data set for this question. Assume the mean kilojoule content of a fast food meal is 2,000 KJ, and that the associated standard deviation is 220 KJ. The researcher...
-
From what alkyl bromide was the following alkyl acetate made by SN2 reaction? Write the reaction, showing allstereochemistry.
-
Draw the structure and assign Z or F stereochemistry to the product you expect from E2 reaction of the following molecule with NaOH (yellow-green =Cl):
-
How can the NCAA enforce its cartel without the explicit power of the government behind it? How can the major leagues enforce their caps?
-
A 34-year-old male certified public accountant, has struggledwith being overweight since early childhood. His currentanthropometric measurements are: 61, 427 pounds. After months ofcareful...
-
You are taking out a car loan for $19,000, and you are given the options below. Find the monthly payment and the total amount of money you will pay for each loan in dollars. (Round your answers to...
-
A 250-pound (113.6-kg) football player participates in a twohour morning practice in warm conditions. Following the workout,the athletic trainer measures the players post practice bodyweight as 245...
-
3. Black-Scholes Option Pricing Assume that a stock price follows a geometric Brownian motion with a drift of 15% and a volatility of 30%. The current stock price is $50. (a) What is the probability...
-
Matt Simpson owns and operates Quality Craft Rentals, which offers canoe rentals and shuttle service on the Nantahala River. Customers can rent canoes at one station, enter the river there, and exit...
-
For the following data set: a. Construct the multiple regression equation b. Predict the value of y when x1 = 10.1, x2 = 8.5, x3 = 26.2. c. What percentage of the variation in y is explained by the...
-
Rosalie owns 50% of the outstanding stock of Salmon Corporation. In a qualifying stock redemption, Salmon distributes $80,000 to Rosalie in exchange for one-half of her shares, which have a basis of...
-
Consider the reaction: If a solution initially contains 0.210 M HC 2 H 3 O 2 , what is the equilibrium concentration of H 3 O + at 25 C? HCH3O (aq) + HO(1) H3O+(aq) + CH3O (aq) K 1.8 x 10-5 at 25 C =
-
Draw all four resonance forms of the fragment at m/z 73 in the mass spectrum of pentanoic acid.
-
Name the following carboxylic acid derivatives, giving both a common name and an IUPAC name where possible. (a) PhCOOCH2CH(CH3)2 (b) PhOCHO (c) PhCH(CH3) COOCH3 (d) PhNHCOCH2CH(CH3)2 (e) CH3CONHCH2Ph...
-
(a) Show how you would use acetic anhydride and an appropriate alcohol or amine to synthesize (i) benzyl acetate, (ii) N,N-diethylacetamide. (b) Propose a mechanism for each synthesis in part (a).
-
When capturing and documenting system audit requirements for an IT project, the teams is focused on capturing the following
-
Gabriela is a claims adjuster who is conducting a phone interview with a claimant. She has asked a question to openly address an issue and clarify one of the claimant's statements. Gabriella has...
-
In the course, we understood that there are certain key financial reports for the healthcare services organization. The Balance Sheet, Operating Statement (Income Statement; Revenue/Expense...

Study smarter with the SolutionInn App


