At 20?C. 1,000 kg of a mixture of 50 wt% Na2S04 . 10H2O and 50 wt% Na2S04
Question:
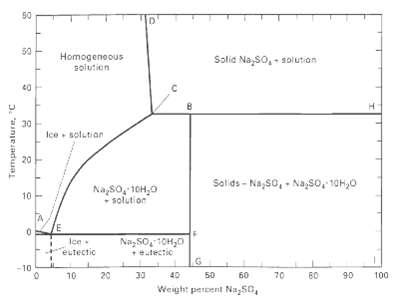
At 20?C. 1,000 kg of a mixture of 50 wt% Na2S04 . 10H2O and 50 wt% Na2S04 crystals exists. How many kilograms of water must be added to just completely dissolve the crystals if the temperature is kept at 20°C and equilibrium is maintained? UseFigure.
Transcribed Image Text:
50 50 Homogeneous solution Solia Na, so,- solution Ice solutian 20 Salids - Na,S0, - Na,So, 10H,0 Na,8o, 10H,0 solution 10 Na, SO, 10H,0 Ice eutectic +ealectio -10 60 10 20 30 40 50 60 70 90 100 Weight percent Na,50, Temperalure, "C 30
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
From Fig the solubility of Na 2 SO 4 in water at 20 o C is 15 wt ...View the full answer

Answered By

Jeff Omollo
As an educator I have had the opportunity to work with students of all ages and backgrounds. Throughout my career, I have developed a teaching style that encourages student engagement and promotes active learning. My education and tutoring skills has enabled me to empower students to become lifelong learners.
5.00+
5+ Reviews
50+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
How many kilograms of nickel must be added to 1.75 kg of copper to yield a liquids temperature of 1300(C?
-
How many kilograms of nickel must be added to 5.43 kg of copper to yield a solidus temperature of 1200(C?
-
How many grams of MgO must be added to 1 kg of NiO to produce a ceramic that has a solidus temperature of 2200 C?
-
Why is it likely to have preexisting normal faults in an orogenic belt?
-
Tarika Ltd. is a profitable small business. It has not, however, given much consideration to internal control. For example, in an attempt to keep clerical and office expenses to a minimum, the...
-
According to the US Bureau of Labor Statistics (BLS), the median weekly earnings of fulltime wage and salary workers by age for the third quarter of 1998 is given in Table 1.4.4. Construct a pie...
-
Suppose you want to buy a house five years from today. You currently have $25,000 saved toward this purchase, but don't think you'll be able to save any more money, other than the interest that the...
-
A violin string has a length of 0.350 m and is tuned to concert G, with fG = 392 Hz. Where must the violinist place her finger to play concert A, with fA = 440 Hz? If this position is to remain...
-
6. Find the foci for (x-6)2 + 9 (y-7)2 25 = = 1 (a) F = (6,11) and F = (6,3) (b) F = (6, 12) and F = (6, 2) (c) F = (-6, 11) and F = (6,3) (d) F = (11,7) and F = (1,7)
-
CC Car Wash specializes in car cleaning services. The ser- vices offered by the company, the exact service time, and the resources needed for each of them are described in the table following: The...
-
Repeat Example 4.11, except let the original solution be 20 wt% Na2S04.
-
Repeat Example 4.12, except determine the grams of activated carbon to achieve: (a) 75% adsorption of phenol. (b) 90% adsorption of phenol. (c) 98% adsorption of phenol.
-
Suppose that we divide commodities into two groups: food and nonfood. (a) Draw indifference curves that reflect the fact that preferences for food grow relatively weaker as we increase consumption of...
-
What is the objective of the Post-Mission Phase of Operation? How do we bound its starting and ending points?
-
Identify the causes of changes to projects, and how to control those changes.
-
Who is responsible for conducting technical reviews?
-
What is the objective of the Mission Phase of Operation? How do we bound its starting and ending points?
-
What is the objective of the Pre-Mission Phase of Operation? How do we bound its starting and ending points?
-
Explain why an estimator with a breakdown point of \(50 \%\) may not give satisfactory results in fitting a regression model.
-
In the synthesis of the keto acid just given, the dicarboxylic acid decarboxylates in a specific way; it gives Explain. HO rather than HO
-
Explain what is meant by push and pull factors in relation to turnover. What can an organization do, if anything, to control these factors?
-
What is diafiltration? How does it differ from continuous feed-and-bleed ultrafiltration? Under what conditions is diafiltration used in conjunction with continuous feed-and-bleed ultrafiltration?
-
In Example 8.19, the % extraction of lysozyme is high (95.2%), but the % purity of the lysozyme in the total extracted proteins is low (15.1%). The Kremser plot of Figure 5.9 suggests that by...
-
What is continuous feed-and-bleed ultrafiltration? What are its limitations?
-
Logistics Solutions provides order fulfillment services for dot.com merchants. The company maintains warehouses that stock items carried by its dot.com clients. When a client receives an order from a...
-
A survey of 150 students who exercise regulariy found that 60 jog, 60 swim, 50 cycle, 15 jog and swim, 5 jogs and cycles, 10 swim and cycle, and 5 does all three. How many of the studenits do not do...
-
DC Electronics uses a standard part in the manufacture of several of its radios. The total cost of producing 30,000 parts is $90,000, which includes fixed costs of $57,000 and variable costs of...

Study smarter with the SolutionInn App


