Calculate the formal charges on the atoms shown inred. (a) (CH3)2OBF3 (b) H2C-NEN: (c) H2C=N=N: (d) :=-:
Question:
Calculate the formal charges on the atoms shown inred.
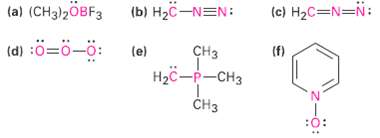
Transcribed Image Text:
(a) (CH3)2OBF3 (b) H2C-NEN: (c) H2C=N=N: (d) :ö=ö-ö: (e) сHз (f) Нас —р-снз CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
To save space molecules are shown as linebond structures with lone pairs rather than as electrondo...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Calculate the formal charges on all of the atoms except hydrogen's, in these compounds: a) H-N-N=N: c) H-C-N=N: H e) H H-=C-H b) H-N-N-N: HO: H-C-C-C 1 H d) f) H H-B-H T H
-
Calculate the formal charges on each of the atoms, except hydrogen's, of the molecules and determine the total charge of the species: a) H H-C-0_H d) H H C=N=N: b) H H-C-0: f) SF: : | H-NC-C- ...
-
Verify that the formal charges on nitrogen in ammonium ion and boron in borohydride ion are as shown.
-
There are 38 numbers in the game of roulette. They are 00, 0, 1, 2, . . ., 36. Each number has an equal chance of being selected. In the game, the winning number is found by a spin of the wheel. Say...
-
Ricky owns stock in Dove Corporation. His adjusted basis for the stock is $90,000. During the year, he receives a distribution from the corporation of $75,000 that is labeled a return of capital...
-
On April 12, 1955, Dr. Jonas Salk released the results of clinical trials for his vaccine to prevent polio. In these clinical trials, 400,000 children were randomly divided in two groups. The...
-
Electronix Plus manufactures private-label small electronic products, such as alarm clocks, calculators, kitchen timers, stopwatches, and automatic pencil sharpeners. Some of the products are sold as...
-
As CEO of Riverside Marine, Rachel Moore knows it is important to control costs and to respond quickly to changes in the highly competitive boat-building industry. When Gerbig Consulting proposes...
-
Acme Company Balance Sheet As of January 5, 2022 (amounts in thousands) Cash 12,500 Accounts Payable 2,400 Accounts Receivable 3,600 Debt 3,700 Inventory 5,200 Other Liabilities 3,000 Property Plant...
-
The Foxridge Investment Group buys and sells rental income properties in southwest Virginia. Bill Hunter, president of Foxridge, has asked for your assistance in analyzing a small apartment building...
-
Methanethiol, CH2SH, has a substantial dipole moment ( = 1.52) even though carbon and sulfur have identical electro negativities. Explain.
-
Which of the following pairs of structures represent resonanceforms? (b) :0: (a) :0: and and (d) :: (c) :0: :0: and and
-
Imagine that youre the owner of a retail store and decide to close three of your 14 locations. Working in groups, identify in the communication plan template in Figure 14 which medium you would use...
-
Oriole Farm Supply Company manufactures and sells a pesticide called Snare. The following data are available for preparing budgets for Snare for the first 2 quarters of 2025. 1. Sales: quarter...
-
Why are references important in research?
-
2. Depreciation On July 1, 2022 your company purchased a vehicle for $42,500. You expect to use it for 4 years or 100,000 km, at which point you will sell it for $2,500. Calculate and journalize...
-
Marta Communications, Inc. has provided incomplete financial statements for the month ended March 3 1 . The Controller has asked you to help calculate the missing amounts in the incomplete financial...
-
Required: 1. Compute Pittman Company's break-even point in dollar sales for next year assuming: a. The agents' commission rate remains unchanged at 15%. b. The agents' commission rate is increased to...
-
Which journal columns are used to record paying cash for insurance?
-
Question 6.10 Current and deferred tax worksheets and tax entries From the hip Ltd?s statement of profit or loss for the year ended 30 June 2007 and extracts from its statements of financial position...
-
The following equation of state has been proposed for a fluid where B and C are constants. a. Does this fluid exhibit a critcal point? Prove it. b. If you believe the answer to part a. is yes, derive...
-
Draw both chair conformations of 1-methyl-1-phenylcyclohexane. Which is more stable by how much energy?
-
Explain whether the methyl is axial or equatorial in this compound: CH3 H C-C-CH3 CH3
-
Draw both chair conformations for menthol (a component of peppermint oil) and its stereo isomer, neo menthol. Which groups are axial and which groups are equatorial? Explain which conformation is...
-
It has been said that the next generation of auditing seeks to take full advantage of emerging technologies in a way that will improve audit quality. Which emerging technology do you believe has the...
-
Which of the following options is not considered a qualified adoption expense under the IRC? A. Expenses incurred that are not reimbursed under another program. B. Expenses directly related to the...
-
Define the Salaries payable account by selecting the appropriate statement below. Multiple choice question. It reports amounts owed to employees and is a liability. It reports amounts owed to...

Study smarter with the SolutionInn App


