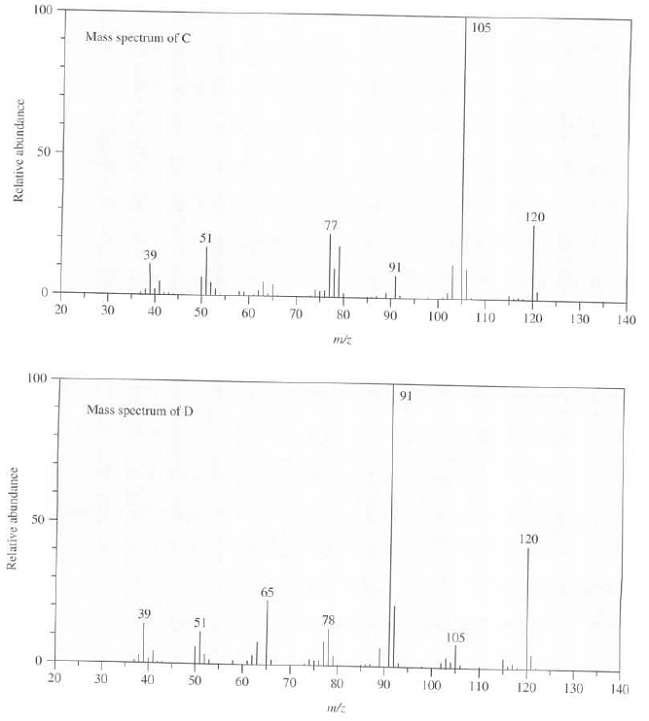
Compounds C and D are isomers with the formula C9H12, in addition to other absorption peaks, both
Question:
Compounds C and D are isomers with the formula C9H12, in addition to other absorption peaks, both compounds show a peak near 7.25 ? (area 5) in their 1H-NMR spectra. Their mass spectra are as follow, show the structures of C and D.
Transcribed Image Text:
100 105 Mass spectrum of C 50 120 77 31 39 91 20 30 40 50 70 60 120 80 90 100 110 130 140 100 91 Mass spectrum of D 50 120 65 39 78 51 105 0. 20 30 40 50 60 70 80 90 100 110 120 130 140 Relative abundance Relative abundance
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
The isomers have DU 4 The 1 HNMR peak near 725 area 5 indicates the presence ...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Compounds A, B, and C are isomers with the formula C5H11Br. Their broadband proton-decoupled 13C NMR spectra are given in Fig. 9.32. Information from the DEPT 13C NMR spectra is given near each peak....
-
Compounds A and B are isomers with the formula C3H6O. A has a peak at 1730 cm ?1 in its IR spectrum and B has a peak at 1715cm ?1 , the mass spectra of A and B are as follows, show the structures of...
-
Compounds Y and Z are isomers with the molecular formula C10H12O. The IR spectrum of each compound shows a strong absorption band near 1710 cm-1. The 1H NMR spectra of Y and Z are given in Figs. 16.4...
-
Write a check (Figure 4 - 42) dated June 13, 20XX, to Byron Johnson in the amount of $296.83 for a washing machine, Complete the check stub. Figure 4 - 42 456 456 Amount To For KRA, INC 2596 Jason...
-
From what you have learnt so far, briefly explain the key responsibilities of public administrators and the best way of meeting the responsibilities.
-
Each team member is to become an expert on one depreciation method to facilitate teammates understanding of that method. Follow these procedures: a. Each team member is to select an area for...
-
Postponing the project means that cash flows come later rather than sooner; however, waiting may allow you to take advantage of changing conditions. It might make sense, however, to proceed today if...
-
Texcar Co. is considering the following alternative financing plans: Income tax is estimated at 40% of income. Determine the earnings per share of common stock, assuming income before bond interest...
-
Perform a vertical analysis on the following information. Note: Round your answers to 1 decimal place.
-
You meet with a prospective client, Tamako, the vice president of human resources for a software development firm. She would like to implement a group savings and investment plan for her employees...
-
The mass spectra of 3-ethyl-2-pentanone and 4-methyl-2-pentanone are as follows. Explain which spectrum goes with which compound, what is the structure of the ion responsible for the peak at m/z 43...
-
Compounds E and F are isomers with the formula C6H8 both react with H2 in the presence of Pt to give G (C6H12), G shows a single peak in its 13C-NMR spectrum. E has no absorption maximum above 200nm...
-
Record merchandising transactions: perpetual inventory system. (LO 1, 2) Assume the following transactions for Jennifer's Fix-It-Up Inc. took place during March. Jennifer's uses a perpetual inventory...
-
Do you think you would be good at babysitting egos? Think you can handle the fickle media, long hours, and first-class seats on airplanes and at sporting events? Then become a sport agentits good...
-
Practice writing a press release for a local sporting team. You might also write a press release for a fantasy sport team (if you are part of a fantasy league) you manage.
-
So, you still think being an athletic director for a major university is an easy job? Think again. For instance, scheduling games in all sports and at all divisions has become increasingly more...
-
What athletes in the past 6 months have been punished as a way to discourage a certain behavior? What was their punishment?
-
Which situational leadership model do you prefer? Why? Will you use it? Why or why not?
-
EX 9.10 Describe the role of the Alert class.
-
When you weigh yourself on good old terra firma (solid ground), your weight is 142 lb. In an elevator your apparent weight is 121 lb. What are the direction and magnitude of the elevator's...
-
Some policymakers in the United States have argued in favor of eliminating $100 bills in order to make large cash transactions undertaken by drug dealers more difficult. Could the former Soviet...
-
State whether you would expect the entropy change, So, to be positive, negative, or approximately zero for each of the following reactions. (Assume the reactions take place in the gas phase.) (a) A +...
-
(a) What is the value of Go for a reaction where Keq = 1? (b) Where Keq = 10? (The change in Go required to produce a 10-fold increase in the equilibrium constant is a useful term to remember.) (c)...
-
Draw contributing resonance structures and a hybrid resonance structure that explain two related facts: the carbon-oxygen bond distances in the acetate ion are the same, and the oxygen's of the...
-
Consider the following data for Microsoft: Selling price per unit: $100 Variable cost per unit: $60 Fixed costs: $300,000 i) Calculate the contribution margin per unit. ii) Determine the break-even...
-
Marginal Cost Analysis at Toyota Values: Variable Cost per Unit: $50 Selling Price per Unit: $80 Fixed Costs: $500,000 Requirements: Calculate the contribution margin per unit and ratio. Determine...
-
Standard Costing at Ford Values: Cost Element Standard Cost Actual Cost Material Cost $15 per unit $14.50 per unit Labor Cost $35 per hour $37 per hour Requirements: Calculate the material price...

Study smarter with the SolutionInn App


