Consider a large furnace with opaque, diffuse, gray walls at 3000 K having an emissivity of 0.85.
Question:
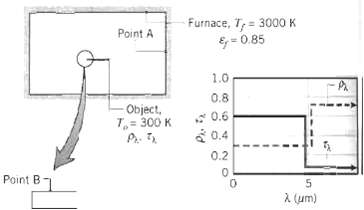
Consider a large furnace with opaque, diffuse, gray walls at 3000 K having an emissivity of 0.85. A small, diffuse, spectrally selective object in the furnace is maintained at 300 K. For the specified points on the furnace wall (A) and the object (B), indicate values for ?, ?, E, G, and J.
Transcribed Image Text:
Furnace, T,= 3000 K Point A E; = 0.85 1.0 0.8 Object, 7,= 300 K * 0.6 a 0.4 0.2 Point B A (um)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 68% (16 reviews)
KNOWN Large furnace with diffuse opaque walls T f and a small diffuse spectrally selective object To ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Heat and Mass Transfer
ISBN: 978-0471457282
6th Edition
Authors: Incropera, Dewitt, Bergman, Lavine
Question Posted:
Students also viewed these Mechanical Engineering questions
-
A small gray sphere having an emissivity of 0.5 and a surface temperature of 1000F is located in a blackbody enclosure having a temperature of 100F. Calculate for this system: (a) The net rate of...
-
The spectral emissivity of an opaque surface at 1500 K is approximated as Determine the total emissivity and the emissive flux of thesurface. for <2 m | -0 0.85 ) for 2 6 for for >6 =0
-
A furnace wall is to be constructed of brick having standard dimensions 9 by 4.5 by 3 in. Two kinds of material are available. One has a maximum usable temperature of 1900?F and a thermal...
-
O 00:29:33 4. Let an be a convergent series and b, be a soquence such that 0 <2+ a, < b Which of the following statements are true? 10 I) The convergence or divergence of cannot be concluded. Im (2+...
-
When asked what business schools should teach more or less of, venture capitalist Guy Kawasaki said, "They should teach students how to communicate in five-sentence e-mails and with 10-slide...
-
Consider the high school SAT scores data from Exercise 31. In exercise a) Find a 90% confidence interval for the slope of the true line describing the association between Math and Verbal scores. b)...
-
If the sale were denominated in U.S. dollars at the April 1 exchange rate, what would be the journal entries for parts (a) and (b) of Exercise 3 in pounds sterling for the British importer?
-
Buffalo Wild Wings, Inc. owns and operates more than 550 Buffalo Wild Wings, R Taco, and PizzaRev restaurants. Additionally, the corporation has franchised restaurants in both the United States and...
-
f(x)=3+5x+7, and f(3) = 49. Let g(x) be the inverse function of f(x). g'(49) Write the equation of the tangent line to g(x) at x = 45
-
In this chapter, we identified the method of authority, the rational method, and the empirical method as techniques for acquiring information. For each of the following, choose one of these three...
-
The spectral absorptivity ?? and spectral reflectivity p? for a spectrally selective, diffuse material are as shown. (a) Sketch the spectral transmissivity ??. (b) If solar irradiation with GS = 750...
-
Four diffuse surfaces having the spectral characteristics shown are at 300 K and are exposed to solar radiation, which of the surfaces may be approximated as being gray? 0.8 0.8 Surtace A Surface B...
-
Borane (BH 3 ) is very unstable and quite reactive. Draw a Lewis structure of borane and explain the source of the instability.
-
In April, Mr. Jones entered into a contract with a farmer to buy 1000 bushels of peaches for $5,000 (or $5 per bushel). The contract specifies that the peaches must be delivered by August 1. (Mr....
-
you to play the role of an international marketing manager from an international consulting company. Your mission is helping your selected Japanese company in Japan to marketing its product or...
-
Is personal spending linearly related to orders for durable goods and personal income? A recent study reported the amounts of personal spending (in trillions of dollars), amount spent on durable...
-
How can leaving or separation in an organization, whether by choice or not, be handled as smoothly as possible by HR? Explain.
-
On March 1, 2021, Navy Corporation used excess cash to purchase U.S. Treasury bonds for $109,000 plus accrued interest. The bonds were purchased at face value. The appropriate interest rate is 6%....
-
The trial balance for Gold Rush Exploration Company, Inc. does not balance. The following errors were detected: a. The cash, balance is overstated by \($2,000\). b. Rent expense of \($390\) was...
-
Write the expression in radical notation. Then evaluate the expression when the result is an integer. 23 -1/2
-
Which is the correct Lewis structure for nitrogen trifluoride? a) :F=N-F: F: c) :F-N-F: :F: b) :F-N-F: F: d) :F-N-F-F:
-
In Fig. P2.12 the tank contains water and immiscible oil at 20°C. What is h in centimeters if the density of the oil is 898 kg/m3? 6 cm 12 cmOil Water 8 cm
-
In Fig P2.13 the 20°C water and gasoline are open to the atmosphere and are at the same elevation. What is the height h in the third liquid? 1.5 matergasoline 1 mLiquid, SG 1.60
-
The closed tank in Fig P2.14 is at 20°C. If the pressure at A is 95 kPa absolute, determine p at B (absolute). What percent error do you make by neglecting the specific weight of the air? air air...
-
es Pension data for Sterling Properties include the following: Service cost, 2021 Projected benefit obligation, January 1, 2021 Plan assets (fair value), January 1, 2021 Prior service cost-AOCI (2021...
-
Weaver Corporation had the following stock issued and outstanding at January 1, Year 1. 1. 114,000 shares of $7 par common stock. 2. 9,500 shares of $100 par, 7 percent, noncumulative preferred...
-
Prepare year-end adjustments for the following transactions. Omit explanations. 1. Accrued interest on notes receivable is $105. 2. Unearned revenues earned totals $2,200. 3. Eighteen months rent,...

Study smarter with the SolutionInn App


