Develop the feasible product composition regions for the system of figure if the feed composition is 50
Question:
Develop the feasible product composition regions for the system of figure if the feed composition is 50 mol% chloroform, 25 mol% methanol, and 25 mol%acetone.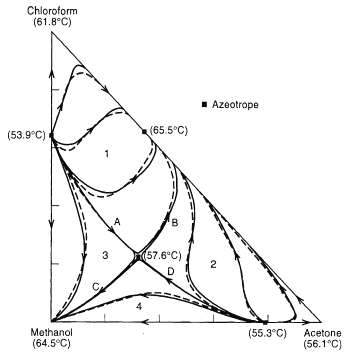
Transcribed Image Text:
Chloroform (61.8°C) 1 Azootrope (65.5°C) (53.9°C) (57.6°C) 3 D. Methanol (55.3°C) Acetone (64.5°C) (56.1°C) B.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
The feed composition is shown at F Thus the feed is in Region 1 which is bounded by distillation bou...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Chemical Engineering questions
-
Develop the feasible product-composition regions for the system of figure, using FeedF1. D2 D, (b) B, Region Region 2 (a) B3 B.
-
For the system of figure below find the transfer function G(s)=X 1 (s)/F(s). K1 = 4 N/m K2 = 5 N/m f(t) fy, = 3 N-s/m M =Ikg Jv2=3N-s/m M,= 2 kg fva = 2 N-s/m! %3D
-
For the system of Figure 1, what happens to the two cylinders in each case? a. Push-button 1-PB is momentarily depressed. b. Push-button 2-PB is momentarily depressed. Note that cylinder 2 does not...
-
Oliveras Company had net credit sales during the year of $800,000 and cost of goods sold of $500,000. The balance in accounts receivable at the beginning of the year was $100,000, and the end of the...
-
A $110,000 payment is made on a long-term liability. Of this amount, $10,000 represents interest. Which of the following is not true for the recording of this transaction? a. Reduce liabilities by...
-
Chicago Tool and Die Corporation (CTDC) recently purchased an aircraft for the top executives to use for customer visits, transporting employees from one location to another, and matters relating to...
-
Suppose you bought a bond that will pay \($1,000\) in 20 years. No intermediate coupon payments will be made. If the appropriate discount rate for the bond is 8 percent, a. what is the current price...
-
Rojas Corporations comparative balance sheets are presented below. Additional information: 1. Net income was $22,630. Dividends declared and paid were $19,500. 2. All other changes in noncurrent...
-
[The following information applies to the questions displayed below.] The stockholders' equity section of TVX Company on February 4 follows. Common stock-$10 par value, 150,000 shares authorized,...
-
Veterinary clinic Happy Pets is a veterinary clinic operating in Auckland. Recently John Tilley, the manager of the clinic, has been concerned about cash flow shortages, which arose quite...
-
For the same conditions as Exercise 11.5, use a process-simulation program with the UNIFAC equation to calculate a portion of a distillation curve at 1 atm.
-
Repeat Example 11.3, but with ethanol as the solvent.
-
An investment costs $23,958 and will generate cash flow of $6,000 annually for five years. The firms cost of capital is 10 percent. a. What is the investments internal rate of return? Based on the...
-
Based on the data in the video (100 pages for $5.25, 200 pages for $7.75, 300 pages for $10.25, 400 pages for $12.75), a) How well will a line model the data? Select an answer b) Find an equation for...
-
An investor purchased a stock three years ago for $15 and just sold it for $25. What average annual geometric return did she earn?
-
How much is $120,000 worth ten years from now if the interest rate, applied monthly, is 4.5%
-
Budgeted unit sales 1st Quarter 2nd Quarter 3rd Quarter 4th Quarter 15,000 16,000 14,000 13,000 The company's variable selling and administrative expense per unit is $2.50. Fixed selling and...
-
Solve by the addition property of equality. t-24 = -21 If needed, write as a reduced fraction.
-
Describe the role and importance of distinctive competence in international strategy formulation.
-
A simple random sample of 220 university students were asked what pasta they usually order and with which sauce. The preferences of these respondents are summarised below: Sauce Bolognese Pasta...
-
List three factors that can cause a financial crisis.
-
Fifty thousand lb/h of a 20 wt% aqueous solution of NaOH at 120F is to be fed to an evaporator operating at 3.7 psia, where the solution is concentrated to 40 wt% NaOH. The heating medium is...
-
A desublimation unit of the heat-exchanger type is to be sized for the recovery of 200 kg/h of benzoic acid (BA) from a gas stream containing 0.8 mol% BA and 99.2 mol% N 2 . The gas enters the unit...
-
A bar of 98 wt% Al with 2 wt% of Fe impurity is subjected to one pass of zone refining. The solidliquid distribution coefficient for the impurity is 0.29. If z/l = 10 and the resulting bar is cut off...
-
This question: 1 point(s) possible What is the effect on the equilibrium price and equilibrium quantity of magazines if magazines become less popular and a more productive printingtechnolog The...
-
K Monopolistic competition is similar to perfect competition because OA. in both industry structures, the firm's demand curve is horizontal. B. both industry structures have a single seller. OC. in...
-
Select the correct answer. GoGo Gas and Fab Fuel are among the few large producers of gasoline in the country. Along with other large producers, they have formed a formal organization to control the...

Study smarter with the SolutionInn App


