Draw the important resonance structures for these species and discuss the contribution of each to the resonance
Question:
Draw the important resonance structures for these species and discuss the contribution of each to the resonance hybrid. Explain whether the species has a large or a small amount of resonance stabilization?
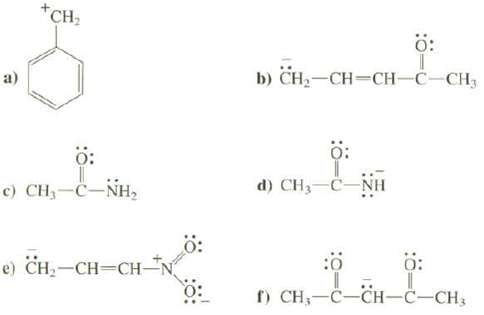
Transcribed Image Text:
a) +CH₂ Ö: c) CH,—C−NH, e) CH₂-CH=CH- b) CH₂-CH=CH-C-CH3 Ö: d) CH₂-C-NH Ö: :0 Ö: f) CH-C-CH-C-CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
a All of these resonance structures are important Although the carbocation is unstable because it do...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Draw the important resonance structures for these species. Use the curved arrow convention to show how the electrons are moved to create each new resonance structure. Discuss the relative...
-
For each of these ions, draw the important resonance forms and predict which resonance form is likely to be the major contributor. (a) (b) (c) CH2 CH CH2 HN
-
Show the important resonance structures for these species. Use the curved arrow convention to show how the electrons are moved to create each new resonance structure. ) d) . CH3 H- b) CH3-N + H -H...
-
Taxpayer sold the following capital assets during the year: Asset # 1: $60,000 long-term capital gain Asset # 2: $10,000 long-term capital loss Asset # 3: $50,000 short-term capital gain Asset # 4:...
-
Explain how the terms controllability and attributability are relevant when evaluating the performance of business unit managers and business units?
-
Minelli Enterprises uses large amounts of copper in the manufacture of ceiling fans. The firm has been very concerned about the detrimental impact of rising copper prices on its earnings and has...
-
On September 24 MJ Foods of Chicago sold \(\$ 2,400,000\) of corn oil to Taj Bakeries, Ltd. of Madras, India, payable in three months. The sale was denominated in Indian rupees at a time when the...
-
Competition for a new generation of computers is so intense that Micro Tech has funded three separate design teams to create the new systems. Due to varying capabilities of the team members, it is...
-
Some researchers developing a new intelligence test are trying to decide how much time to allow to complete the test. The researchers have recorded the times (in minutes) for completion of 29 people...
-
There is a lottery with n coupons and n people take part in it. Each person picks exactly one coupon. Coupons are numbered consecutively from 1 to n, n being the maximum ticket number. The winner of...
-
In these examples the additional structure or structures are not important contributors to the resonance hybrid for the compound represented by the first structure, explain. a) 8-8 c) CH-C=N: b) :0...
-
Explain why this carbocation is considerably more stable than this structure would suggest: H +C-0-CH, H
-
Urea, used in the experiment shown in Figure 47, is a molecule that disrupts the hydrogen-bonded network of water molecules. Why might high concentrations of urea unfold proteins? The structure of...
-
a bathroom scale could also be used to determine the gravitational field strength. An 80kg astronaut is working on the surface of a small asteroid and steps on a bathroom scale. If the scale reads...
-
What is the formula for calculating profit margin? Explain the meaning of each variable used in the calculation
-
Give fifteen (15) risks/problems encountered by credit cooperatives on collection of loans/payments.
-
1) A battery of 5 V is hooked up to a resistor of 10. What is the current? 2) A battery is hooked up to a resistor of 30 2 and the ammeter reads a current of 0.2 A. What is the voltage of the...
-
The following data has been provided by Lee Company regarding its inventory purchases and sales throughout the year. Transaction Units Cost per Unit January 1 Balance 185 $86 March 14 Sale 54 May 23...
-
Compare and contrast the three stages of functional aggregation.
-
Air pollution generated by a steel mill is an example of a) a positive production externality. b) a negative production externality. c) a public good. d) the free-rider problem. State and local taxes...
-
For what values of p is the series convergent? n=1 1 nP
-
Malic acid, C4H6O5, has been isolated from apples. Because this compound reacts with 2 molar equivalents of base, it is a dicarboxylic acid. (a) Draw at least five possible structures. (b) If malic...
-
Formaldehyde, H2C = O, is known to all biologists because of its usefulness as a tissue preservative. When pure, formaldehyde trimerizes to give trioxane, C3H6O3, which, surprisingly enough, has no...
-
Increased substitution around a bond leads to increased strain. Take the four substituted butanes listed below, for example. For each compound, sight along the C2-C3 bond and draw Newman projections...
-
With respect to the adjustments made by the analyst in Country B related to property, plant, and equipment, answer the following: 1. Why does the adjustment for depreciation related to revalued...
-
Yolanda tiene una tienda de productos de computacin, su objetivo principal es poder mantener econmicamente a su familia, pues la venta de computadoras es un buen negocio. La situacin mostrada de...
-
The income statement of Novak Co. for the month of July shows net income of 3,280 based on Service Revenue 7,750, Salaries and Wages Expense 2,920, supplies expense 930, and Utilities Expenses 620....

Study smarter with the SolutionInn App


