Draw the two chair conformations of menthol, and tell which is morestable. C Menthol CH(CH3)2
Question:
Draw the two chair conformations of menthol, and tell which is morestable.
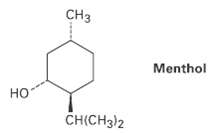
Transcribed Image Text:
Cнз Menthol но CH(CH3)2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
From the flatring drawing you can see that the methyl group and the OH group have a cis relation...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Draw the two chair conformations of the sugar α-(+)-glucopyranose, one form of the sugar glucose. Which of these two forms is the major one at equilibrium? Explain. CH2OH OH ...
-
Draw the two chair conformations of cis-1-chloro-2-methylcyclohexane. Which is more stable, and by how much?
-
Draw the two chair conformations of trans-1-chloro-2-mcthylcyclohexane. Which is more stable?
-
A strange function. Consider McCarthys 91 function: public static int mcCarthy(int n) { if (n > 100) return n - 10; return mcCarthy(mcCarthy(n+11)); } Determine the value of mcCarthy(50) without...
-
Tammy Jackson purchased 100 shares of All-American Manufacturing Company stock at $31.50 a share. One year later, she sold the stock for $38 a share. She paid her broker a $15 commission when she...
-
Describe the effect on bank reserves when the Federal Reserve sells U.S. government securities to a bank.
-
5. H. B. Fuller Co. of St. Paul is a leading manufacturer of industrial glues. Its mission statement says the company "will conduct business legally and ethically." It has endowed a university chair...
-
You have been asked to audit Greystone Company. During the course of your audit, you are asked to prepare comparative data from the company's inception to the present. You have determined the...
-
can you please assist with answer. On 30 June 2021, Cold Ltd determined that due to a downturn in the market for DVD players that impairment testing needed to be conducted on the assets held in the...
-
Consider the demand for Fresh Detergent in a future sales period when Enterprise Industries' price for Fresh will be x1 = 3.70, the average price of competitors" similar detergents will be x2 = 3.90,...
-
Galactose, a sugar related to glucose, contains a six-membered ring in which all the substituents except the ?OH group indicated below in red are equatorial. Draw galactose in its more stable chair...
-
There are four cistransisomers of menthol (Problem 4.37), including the one shown. Draw the other three.
-
The following accounts appear in the ledger of Tiger Inc. after the books are closed at December 31. Common Stock, no par, $1 stated value, 400,000 shares authorized; 300,000 shares issued...
-
The Coulomb's law which calculates the force between two point charges q and q2 is given by the following equation, where co=8.854 x 102 C/N-m is the vacuum permittivity or electric constant, r =...
-
With access to the spacecraft's controls, you connect your computer and begin installing the fuel tank and start capacitor. Your computer informs you that it can plot most of the course to your home...
-
We are starting a new company on January 1, 2021. As part of our start-up planning, we are developing a cash budget for 2021: On January 1, we plan to have $250,000 in cash for the launch of the...
-
A rectangular conducting metal loop is being pulled out of an area with a constant magnetic field. The loop has a length L and width W, a resistance R, and is moving with a constant velocity v to the...
-
Carlin Company has current assets of $100,000, total assets of $1,000,000, current liabilities of $50,000, total liabilities of $250,000 and total equity of $750,000. What is the current ratio...
-
Increasingly, householders and small businesses are considering renewable power generation systems to decrease their costs. Most of the UK, Ireland and Scandinavia are suited to generating energy...
-
Discuss whether responsible human resources management should apply different standards for the home company and suppliers, for developed countries and developing countries, and for large companies...
-
Arrange the following substances in the order in which you would expect their boiling points to increase: CCl 4 , Cl 2 , ClNO, N 2 .
-
Provide a mechanism for the following reaction, based on your knowledge of the reaction of esters with Grignard reagents. OH MgBr (2 equiv.) Cl (2) NH,CI
-
Provide retrosynthetic analyses and syntheses for each of the following alcohols, starting with appropriate alkyl or aryl halides. (a) (b) (c) (d) (e) (f) OH (three ways) OH (three ways) (two ways)...
-
Consider the allylic bromination of cyclohexene labeled at C3 with 13C. Neglecting stereoisomers, what products would you expect from this reaction? NBS, ROOR heat (-13C-labeled position)
-
A floating rate note pays quarterly coupons on the 15th of March, June, September, and December of each year based on three-month Market Reference Rate (MRR) + 50 bps. Today is December 15th and the...
-
Which one of these activities represents a source of cash? Select one: a.Decreasing accounts payable b.Increasing accounts receivable c.Decreasing inventory d.Decreasing common stock e.Increasing...
-
Given the formula below calculate the Medical Expense Tax Credit and listed variables, answer the following questions Medical Expense Tax Credit (METC) = ((Eligible Medical Expenses - the lesser of...

Study smarter with the SolutionInn App


