Each of the following syntheses requires more than one step. How would you carry themout? (a) CH3CH2CH2C=CH
Question:
Each of the following syntheses requires more than one step. How would you carry themout?
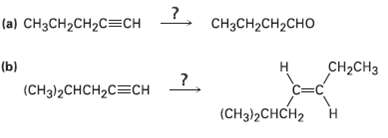
Transcribed Image Text:
(a) CH3CH2CH2C=CH CH3CH2CH2CHO (b) (CHз)2CHCH2CСH CH-CHз н C=C (CHд)2CHCH2 ?. Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (15 reviews)
a b H Lindlar catalyst CH3CHCH...View the full answer

Answered By

Deepak Pal
Hi there! Are you looking for a committed, reliable, and enthusiastic tutor? Well, teaching and learning are more of a second nature to me, having been raised by parents who are both teachers. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students. Areas of interest: Business, accounting, Project management, sociology, technology, computers, English, linguistics, media, philosophy, political science, statistics, data science, Excel, psychology, art, history, health education, gender studies, cultural studies, ethics, religion. I am also decent with math(s) & Programming. If you have a project you think I can take on, please feel welcome to invite me, and I'm going to check it out!
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you carry out the following conversions? More than one step is needed in eachcase. "
-
Syntheses of each of the following compounds have been reported in the chemical literature. Using the indicated starting material and any necessary organic or inorganic reagents, describe short...
-
Syntheses of each of the following compounds have been reported in the chemical literature. Using the indicated starting material and any necessary organic or inorganic reagents, describe short...
-
Spencer Company sells lamps and other lighting fixtures. The purchasing department manager prepared the following inventory purchases budget. Spencers policy is to maintain an ending inventory...
-
In the previous problem, suppose that vendor A provides a new price-discount schedule for component 3. This one is an "incremental" discount, as opposed to an "all-units" discount, as follows: Unit...
-
Exclude leap years from the following calculations: (a) Compute the probability that a randomly selected person does not have a birthday on November 8. (b) Compute the probability that a randomly...
-
1.3 External Debt. The World Bank regularly com- piles and analyzes the external debt of all countries globally. As part of its annual publication on World Development Indicators (WDI), it provides...
-
Turner Company ended its fiscal year on July 31, 2012. The company's adjusted trial balance as of the end of its fiscal year is as shown below. Instructions (a) Prepare the closing entries using page...
-
You are an audit manager in Judd & Co, a firm of Chartered Certified Accountants, and you are working on the audit of Filter Co for the year ended 31 December 2018. Profit for the year is $950,000...
-
ambling was a hot issue in the state of Florida. A survey conducted in 2006 found that 1035 of 1523 Americans supported legalized gambling. Does this survey provide sufficient evidence that more than...
-
Propose structures for hydrocarbons that give the following products on oxidative cleavage by KMnO4 orO3: (b) .Co2 (a) O2 + CH(CH2)5C02H CC2H + e) O2CICH2)8CO2H (d) CH + 2H2cO2H + co2 (e) CO2...
-
How would you carry out the following transformation? More than one step isneeded. CH3CH2CH2CH2C=CH T ? CCH2CH2CH2 H
-
If a client is not deemed to have significant influence in an investee company, to obtain audit evidence to verify the investment balance, the auditor should: (a) review the audited accounts of the...
-
On December 3 1 , 2 0 2 3 , Intermediate CPA Inc., had net income of $ 3 , 1 0 0 , 0 0 0 . During the year, Common Stock were outstanding as follows: January 1 , Beginning 4 0 0 , 0 0 0 , Issued on...
-
Winfrey Designs had an unadjusted credit balance in its Allowance for Doubtful Accounts at December 3 1 , 2 0 2 3 , of $ 2 , 4 0 0 . Required: a . Prepare the adjusting entry assuming that Winfrey...
-
Andretti Company has a single product called a Dak. The company normally produces and sells 8 3 , 0 0 0 Daks each year at a selling price of $ 5 6 per unit. The company s unit costs at this level of...
-
Explain how stocks are valued. Describe the process of putting stocks out for sale to the public Discuss the elements of Common Stock Explain how you calculate the value of Common Stock Discuss the...
-
Question 2 Talam Bhd. wants to purchase machinery worth RM3,400,000. It will be completely obsolete in five years and the estimated salvage value is zero. If the company purchases the machine, it...
-
Consider the circuit shown in Figure P2.1. Let \(R=1 \mathrm{k} \Omega, V_{\gamma}=0.6 \mathrm{~V}\), and \(r_{f}=20 \Omega\). (a) Plot the voltage transfer characteristics \(v_{O}\) versus \(v_{I}\)...
-
You are the newly appointed tax practitioner to complete Emilys tax return and have downloaded the prefill report for Emilys tax return (hint, you can read what a prefill report is here (Links to an...
-
If 123 mL of a 1.1 M glucose solution is diluted to 500.0 mL, what is the molarity of the diluted solution?
-
Compound X, isolated from lanolin (sheep's wool fat), has the pungent aroma of dirty sweatsocks. A careful analysis showed that compound X contains 62.0% carbon and 10.4% hydrogen. No nitrogen or...
-
For each of the following structures, 1. Draw a Lewis structure; fill in any nonbonding electrons. 2. Calculate the formal charge on each atom other than hydrogen. All are electrically neutral except...
-
1. From what you remember of electronegativities, show the direction of the dipole moments of the following bonds. 2. In each case, predict whether the dipole moment is relatively large...
-
Please correct answers** . Exercise 9-25 Budgeting Production and Raw-Material Purchases (LO 9-3, 9-6) Greener Grass Fertilizer Company plans to sell 280,000 units of finished product in July and...
-
Understanding personality traits is the key to predicting behaviour and understanding how situations can influence individuals. With reference to this statement , discuss numerous traits which...
-
Identify 1 health care organization type (e.g., hospital, nursing home, home health agency) to write about for this assessment. Review the Example Factor Analysis Table and then complete the Factor...

Study smarter with the SolutionInn App


