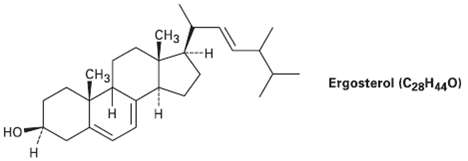
Ergo sterol a precursor of vitamin D, has ? max = 282 nm and molar absorptivity ?
Question:
Ergo sterol a precursor of vitamin D, has ? max = 282 nm and molar absorptivity ? = 11,900. What is the concentration of ergo sterol in a solution whose absorbance A = 0.065 with a sample path length l = 1 .00cm?
Transcribed Image Text:
сНз Ergosterol (C28H440) снз но I.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
c A ex 0065 1...View the full answer

Answered By

AJIN kuriakose
I have completed B.Tech in Electrical Engineering & Masters in Power & Control From one of the best universities in India. I got the 99.05 percentile in the Gate Electrical Engineering Exam. I can Help students solving assignments in Electrical subjects like Power Electronics, Control system, Analog, Network Theory & Engineering Mathematics. Clear your fundamentals and develop problem-solving skills and analytical skills to crack the exam.
Get guidance and the opportunity to learn from experienced...
I can provide tuition for Electrical engineering subjects (Power Electronics, Digital electronics, Network Theory, Control System & Engineering Mathematics). The toughest subject of Electrical engineering can be made simple in online classes...
I can also solve it.
1 .I can help you with your assignments or exams or quiz or tutoring.
2. Very strict to the deadlines.
Message me for any help in assignments, live sessions. I am here to help students for all assignments, tests and exams and I will make sure you always get _95% In your subject.
Contact me in solution inn for any help in your semester, projects and for many more things . Also feel free to contact me through solution inn and for any advise related to tutoring and how it works here.thank you.
5.00+
5+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What is the concentration of hydroxide ion in a 0.060 M aqueous solution of methylamine, CH3NH2? What is the pH?
-
What is the concentration of hydroxide ion in a 0.18 M aqueous solution of hydroxylamine, NH2OH? What is the pH?
-
What is the concentration of hydroxide ion in a 0.21 M aqueous solution of hydroxylamine, NH 2 OH? What is the pH?
-
Sandys Socks makes the worlds best socks. Information for the last eight months follows: Prepare a scatter graph by plotting Sandys data on a graph. Then draw a line that you believe best fits the...
-
If you wanted to hire an accountant to work in New York City, would you search the database for wages in the New York City metropolitan area or for the whole state? Why?
-
Stephanie Baker is an audit senior with the public accounting firm of Wilson & Lang. It is February Year 9, and the audit of Canadian Development Limited (CDL) for the year ended December 31, Year 8,...
-
Percentage Depreciation. Assume the spot rate of the British pound is $1.73. The expected spot rate one year from now is assumed to be $1.66. What percentage depreciation does this reflect?
-
A friend named Jay Barlow has asked what effect certain transactions will have on his company. Time is short, so you cannot apply the detailed procedures of journalizing and posting. Instead, you...
-
1 2 3 4 5 OT 6 7 8 3 of 23 (1 point) Attempt 1 of Unlimited 5 e point 9, A 4 (9. 7/7) 263 in polar coordinates to th
-
The following information pertains to the City of Williamson for 2024, its first year of legal existence. For convenience, assume that all transactions are for the general fund, which has three...
-
Adiponitrile, a starting material used in the manufacture of nylon, can be prepared in three steps from 1, 3-butadiene. How would you carry out thissynthesis? 3 steps H2C=CHCH=CH2 N=CCH2CH2CH2CH2C=N...
-
A 1, 3-Cyclopentadiene polymerizes slowly at room temperature to yield a polymer that has no double bonds except on the ends. On heating, the polymer breaks down to regenerate 1, 3-cyclopentadiene....
-
Search the FASB ASC to find the items classified as current assets. Cite and copy the relevant paragraph.
-
Furniture priced at $20,000 is purchased. A deposit of 15% is paid. The balance is borrowed using a flatrate loan at 19% per annum interest, to be repaid in equal monthly instalments over five years....
-
You have been asked to estimate the cost of capital for the UTX corporation. The company has 7 million shares and 150,000 bonds outstanding at par value $10,000. In addition, it has $300 million in...
-
In a decision in 2022, the U.S. Supreme Court overturned its earlier decision in Roe v. Wade, which had stood for more than 40 years. In Roe v. Wade, the Court had held that the right to choose to...
-
How does this case illustrate clients' cultural differences? Are the views and conduct of the parent in this case the same as you have and would have taken? What ethical obligations does the...
-
Describe the anatomical and physiological characteristics of the renal system, including the nephron structure, renal blood flow regulation, and mechanisms of urine concentration and dilution, and...
-
A _______________ interval estimates the y-value for a particular individual with a given x-value. In Exercises 3 and 4, fill in each blank with the appropriate word or phrase.
-
Provide an example of an aggressive accounting practice. Why is this practice aggressive?
-
The enthalpy of solution for cesium fluoride is -36.8 kJ/mol. What can you conclude about the relative magnitudes of Hsolute and Hhydration ? (a) AHsolute>|AHhydration (b) AHsolute
-
Determine which of the heterocyclic amines just shown are aromatic. Give the reasons for your conclusions.
-
(a) Show how fragmentation occurs to give the base peak at m/z 58 in the mass spectrum of ethyl propyl amine (N-ethylpropan-1- amine), shown below. (b) Show how a similar cleavage in the ethyl group...
-
Propose a mechanism for nitration of pyridine at the 4-position, and show why this orientation is not observed.
-
_______________: The seller agrees to accept a percentage of the sales price and stays on to manage the business for a few more years under the new owner; the remaining portion of the price is...
-
Private, not-for-profit colleges and universities must have a Statement of Financial Position, Statement of Activities, Statement of Cash Flows, and Notes to the Financial Statements included in...
-
You have just been hired into a management position which requires the application of your budgeting skills. You find out that budgeting has not been a priority of the company. You have contacted...

Study smarter with the SolutionInn App


