Explain why one of these anions is much more stable than the other: : a) CH-C-CH-CH b)
Question:
Explain why one of these anions is much more stable than the other:
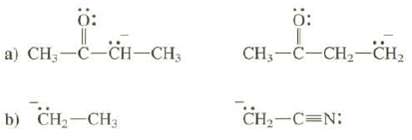
Transcribed Image Text:
Ö: a) CH₂-C-CH-CH₂ b) CH₂ CH3 Ö: CH₂-C-CH₂-CH₂ CH₂-C=N:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
a The anion on the left is more stable because the negative c...View the full answer

Answered By

Lilian Nyambura
Hi, am Lilian Nyambura, With extensive experience in the writing industry, I am the best fit for your writing projects. I am currently pursuing a B.A. in Business Administration. With over 5 years of experience, I can comfortably say I am good in article writing, editing and proofreading, academic writing, resumes and cover letters. I have good command over English grammar, English Basic Skills, English Spelling, English Vocabulary, U.S. English Sentence Structure, U.K. or U.S. English Punctuation and other grammar related topics. Let me help you with all your essays, assignments, projects, dissertations, online exams and other related tasks. Quality is my goal.
4.80+
378+ Reviews
750+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain why one of these carbocations is much more stable than the other: + CH-CH3 CH CH
-
One of the following compounds is much more stable than the other two. Classify each as aromatic, antiaromatic, or nonaromatic. heptalene azulene pentalene
-
Explain why one of these compounds reacts readily by an E2 mechanism when treated with sodium ethoxide in ethanol but the other doesnot: OTs OTS . H, .H, CH; CH3
-
7. A 2.0 kg ball is moving at a velocity of 5.0 m/s. It collides with a stationery ball of mass 2 kg. After the collision the first ball A moves off in a direction 30 to the left of its original...
-
Outline the four purposes of performance measurement systems. Explain how these purposes relate to responsibility accounting?
-
Leading money center banks in the United States have accelerated their investment banking activities all over the globe in recent years, purchasing corporate debt securities and stock from their...
-
Tektronix, Inc., an Oregon-based MNE, manufactures scientific instruments. Jerry Davies, Treasurer, needs to choose an instrument to hedge a \( 2,000,000\) sale to Siemens in Germany, with payment...
-
Cerulean Corporation has two equal shareholders, Eloise and Olivia. Eloise acquired her Cerulean stock three years ago by transferring property worth $700,000, basis of $300,000, for 70 shares of the...
-
13. $16,000 deposit at an APR of 4% with semiannual compounding for 12 years
-
Let Ai be the set of all nonempty bit strings (that is, bit strings of length at least one) of length not exceeding i. Find (a) (b) UA
-
Explain why this carbocation is considerably more stable than this structure would suggest: H +C-0-CH, H
-
Show energy level diagrams for the MOS of these compounds: a) H-C=C-CH3 b) CH0H c) CH3-C-H
-
Explain the purpose and importance of business plan.
-
In the beginning of October, an investor has a stock portfolio worth $3,460,000. The content of the portfolio replicates very closely that of the Shanghai SE A share index. The investor wants to...
-
Suppose you are offered the opportunity to invest with a very popular and active-trading hedge fund. Before investing with that manager, what factors might you consider before exploiting this...
-
Part A A 300-kg chandelier is suspended from vertical steel cable. The cable is 5.7 m long. Assume a safety factor of 7.5. The elastic modulus for steel is 2.0 10 N/m, and tensile strength for steel...
-
Identify the four primary layers in the functional view of OS X ? What is Darwin ? Identify Darwin's two basic layers ? Explain how OS/X thread manages memory ? What is QuickTime ? What does...
-
Swifty Itzek manufactures and sells homemade wine, and he wants to develop a standard cost per gallon. The following are required for production of a 50-gallon batch. 2,880 ounces of grape...
-
What is a horizontal company and how would this type of company be organized? What are the strengths of this type of organization structure?
-
Charles owns an office building and land that are used in his trade or business. The office building and land were acquired in 1978 for $800,000 and $100,000, respectively. During the current year,...
-
(a) What is the maximum error possible in using the approximation when 0.3 x 0.3? Use this approximation to find sin 12 correct to six decimal places. (b) For what values of x is this...
-
Give IUPAC names for the following cycloalkanes: (c) CH2CH2CH3 (b) CH (a) CH CH3 (f) Br (e) C CH-CH (d) CHICH3)2 "CH C(CH3)3 Br
-
Draw structures corresponding to the following IUPAC names: (a) 1, 1-Dimethylcycloocatne (b) 2-Cyclobutylhexane (c) 1, 2-Dichlorocyclopentane (d) 1, 3-Dibromo-5-methylcyclohexane
-
Name the following cycloalkanes:
-
Which of the following statements is correct? Select answer from the options below ASPE permits three measurement models for investments with no significant influence or control; IFRS permits only...
-
top inc acquired all of the outstanding stock of bottom co on january 1, 2020 for $276,000. Annual amortization of $21,000 restulted from this acquisition. Top reported net income of $80,000 in 2020...
-
The head of the accounting department of your company has determined that there is idle cash and is considering options for investing it. You are assigned to explain the costs and benefits of various...

Study smarter with the SolutionInn App


