For each of the ternary systems shown in figure, indicate whether: (a) Simple, countercurrent extraction, or (b)
Question:
For each of the ternary systems shown in figure, indicate whether:
(a) Simple, countercurrent extraction, or
(b) Countercurrent extraction with extract reflux, or
(c) Countercurrent extraction with raffinate reflux, or
(d) Countercurrent extraction with both extract and raffinate reflux would be expected to yield the most economicalprocess.
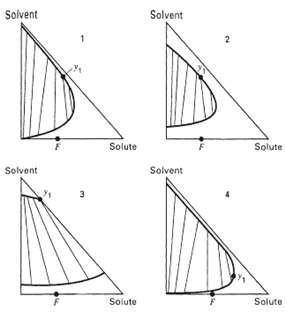
Transcribed Image Text:
Solvent Solvent Solute Solute Solvent Solvent Solute Solute
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (15 reviews)
Subject Selection of extraction method Given Four ternary systems in Figure Diagra...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
For each of the environmental influences shown in Figure 2.1, give examples of why it is important to monitor and respond in an e-business context. For example, the personalization mentioned in the...
-
Indicate the tax treatment for each of the following dividends received by a corporation that is a member of an affiliated group filing a consolidated tax return: a. Dividend received from a...
-
Figure SDifference for each of the following studies: 2222224 20 40 20 40 40 20 20 40 40 40
-
Which of the following variable types can be used in a switch statement under some circumstances? (Choose three.) A. An enumerated type B. StringBuilder C. Byte D. Double E. var F. Exception.
-
During the first month of operations, these events and transactions occurred for Astromech Accounting Services Inc.: May 1 Common shares were issued for $20,000 cash and a $10,000 note receivable. 1...
-
Use the technology instructions provided on the CD to construct the Kaplan- Meier curve for the white and the milk chocolate chips from the chocolate chip melting activity. Compare these two curves....
-
Use PPP to explain how the values of the currencies of Eastern European countries might change if those countries experience high inflation, while the United States experiences low inflation.
-
Discuss the impact, both positive and negative, of termination on the project team members. How might the negative impact be lessened?
-
4. Universal Oil Company obtained the rights to shoot 25,000 acres at a cost of $0.20/acre on May 3, 2007. Universal contracted and paid $80,000 for a reconnaissance survey during 2007. As a result...
-
W&S Partners began the planning phase of the Cloud 9 audit. As part of the risk assessment phase for the new audit, the audit team needs to gain an understanding of Cloud 9's structure and its...
-
The system docosane-diphenylhexane (DPH)-furfural is representative of more complex systems encountered in the solvent refining of lubricating oil. Five hundred kilograms per hour of a 40 wt% mixture...
-
Two solutions, feed F at the rate of 7,500 kg/h containing 50 wt% acetone and 50 wt% water, and feed F' at the rate of 7,500 kg/h containing 25 wt% acetone and 75 wt% water, are to be extracted in a...
-
Kristen, a single taxpayer, receives two 2018 Forms W-2 from the two employers she worked for during the year. One Form W-2 lists her wages in Boxes 1, 3, and 5 as $18,700. Her other employers Form...
-
The audit committee is supposed to be the external auditor's client. Explain what this means and how it is implemented.
-
Does the audit committee need to separately approve nonaudit services to: (a) the company, (b) management, or (c) members of the board of directors? Explain.
-
What does it mean to have management certify that the financial statements are correct? How does this certification differ from the previously acknowledged re- sponsibility for the fairness of the...
-
Sarbanes-Oxley contains certification requirements of management. What are they and how do they differ from the responsibilities that management previously had?
-
What are the whistleblowing provisions of the Sarbanes-Oxley Act? What is the organization's responsibility for establishing and monitoring a Corporate Code of Conduct?
-
State the conditions for the equilibrium of a body under the following system of loading. (a) Two forces, (b) three forces, and (c) two forces and a torque.
-
Quadrilateral EFGH is a kite. Find mG. E H <105 G 50 F
-
A government report forecasts both higher inflation and higher interest rates in the future. Yvette needs to borrow money in six months. What can she, as a future borrower, do now to protect herself...
-
One hundred kmol/h of a three-component bubble-point mixture to be separated by distillation has the composition: (a) For a distillate rate of 60 kmol/h, five stages, and total reflux, calculate the...
-
What is the best method for estimating the distribution of nonkey components at the actual (operating) reflux?
-
The following feed mixture is to be separated by ordinary distillation at 120 psia to obtain 92.5 mol% of the nC 4 in the liquid distillate and 82.0 mol% of the iC 5 in the bottoms. (a) Estimate Nmin...
-
(a) Bonnie contributes land, which she held as inventory (FMV 473,000 and AB 516,000), in exchange for an interest in the JB Partnership. The partnership holds the land as inventory and sells it...
-
Employer health taxes are calculated on: Multiple Choice Calculated on taxable earnings as defined by the jurisdiction T4 employment earnings Earnings up to the annual maximum pensionable earnings...
-
Each tax payment made by most corporations throughout the year a. is a portion of the actual income tax for the previous year. b. is the actual tax on income earned in the previous quarter. c. is a...

Study smarter with the SolutionInn App


