For the ZrO 2 ?CaO system (Figure), write all eutectic and eutectoid reactions for cooling. Composition (mol%
Question:
For the ZrO2?CaO system (Figure), write all eutectic and eutectoid reactions for cooling.
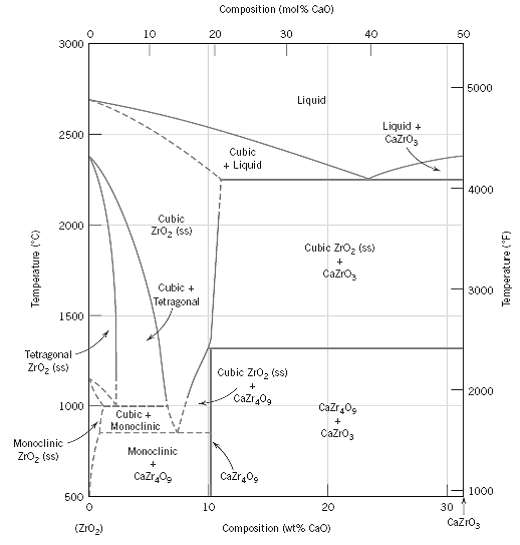
Transcribed Image Text:
Composition (mol% Cao) 10 20 30 40 50 3000 5000 Liquid Liquid + Cazro, 2500 Cubic + Liquid 4000 Cubic 2000 Zro, (ss) Cubic Zro, (ss) Cazro, Cubic + 3000 Tetragonal 1500 Tetragonal Zroz (ss) Cubic ZIO, (ss) 2000 Cazr 0g 1000 Cazr,0g Cubic + Monoclinic Cazro3 Monoclinic Monoclinic ZrOz (ss) Cazr Og Cazr0g 1000 500 10 20 30 CazrO3 Composition (wt% CaO) (Z1O2) (0.) eameredwejL Temperature (F)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
There is only one eutectic for the portion of the ZrO 2 CaO system shown in ...View the full answer

Answered By

Shristi Singh
A freshman year metallurgy and material science student in India.
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
For the ZrO2-CaO system (Figure 12.26), write all eutectic and eutectoid reactions for cooling.
-
Write the eutectic reaction that occurs, including the compositions of the three phases in equilibrium, and calculate the amount of and in the eutectic micro constituent in the Mg-Al system (Figure...
-
Figure 11-32 shows a cooling curve for an Al-Si alloy and Figure 11-19 shows the binary phase diagram for this system. Determine (a) The pouring temperature; (b) The superheat; (c) The liquidus...
-
Reading material Raymond Frost, Alexa K. Fox & Judy Strauss (2019). Product: The Online Offer. E-Marketing , 9, 206 - 228. Raymond Frost, Alexa K. Fox & Judy Strauss (2019). Price: The Online Value....
-
Discuss in depth the demographer of Germany. Outline the discuss by take in to account the following factors Population, financial status, top 3 diseases and overall healthcare, type of healthcare...
-
How does a partnership differ from a corporation?
-
The following questions deal with types of audits and auditors. Select the best response for each question. a. Operational audits generally have been conducted by internal auditors and governmental...
-
Perform analytical procedures for accounts payable of J & J Auto Repair Service in the following manner: a. Calculate and list all necessary figures and comparisons. b. Explain what the result of...
-
5. Precy, Inc., a domestic corporation, reported the following income in 2014. Philippines Abroad Service fees P 400, 000 P 300, 000 Interest income - bank 40 , 000 70, 000 Royalties - franchise 80,...
-
. Venture Inc. is sitting on $100 million in liquidity and considering investing in two different start-ups: Company A and Company B. After 3 years, Venture plans to sell their stake in both...
-
What point defects are possible for Al2O3 as an impurity in MgO? How many Al3+ ions must be added to form each of these defects?
-
From Figure, the phase diagram for the MgO???Al2O3 system, it may be noted that the spinel solid solution exists over a range of compositions, which means that it is nonstoichiometric at compositions...
-
Manuel and Fernando own and operate an electronics store, Electronica, as an S corporation. Manuel owns 70%, and Fernando owns 30%. For the current year, the store reports the following: Sales...
-
_____ provides the maximum, minimum, and midpoint of pay for a certain group of jobs.
-
The balance in the unearned fees account, before adjustment at the end of the year, is \($7,000\). a. Determine the amount of adjustment if the amount of unearned fees at the end of the year is...
-
River Realty Inc. pays weekly salaries of \($10,500\) on Friday for a five-day week ending on that day. a. Determine the amount of the adjustment at the end of the accounting period, assuming that...
-
Jhe balance in the unearned fees account, before adjustment at the end of the year, is \($31,700.\) Of these fees, \($21,500\) have been earned. In addition, \($9,100\) of fees have been earned but...
-
What are the three elements common to most employee fraud?
-
The shaft consists of a solid segment \(A B\) and a hollow segment \(B C\), which are rigidly joined by the coupling at \(B\). If the shaft is made from A-36 steel, determine the maximum torque \(T\)...
-
Where are the olfactory sensory neurons, and why is that site poorly suited for their job?
-
H vap = 31.3 kJ/mol for acetone. If 1.40 kg of water were vaporized to steam in a boiler, how much acetone (in kg) would need to be vaporized to use the same amount of heat?
-
How does a metal increase its internal energy during plastic deformation?
-
In what ways can recrystallization be used to enable large amounts of deformation without fear of fracture?
-
What is the major distinguishing feature between hot and cold working?
-
Why was General Electric chosen to build the Whittle engine? Group of answer choices Due to their experience with turbine engines in aircraft Due to their experience in building reciprocating engines...
-
(8%) Problem 11: The magnitude of a vector is indicated by the length of the arrow representing it. For a vector v, the magnitude can be denoted using vertical bars, such as (v) A True or false: For...
-
Subsonic flights involve incompressible flow of air. Group of answer choices True False

Study smarter with the SolutionInn App


