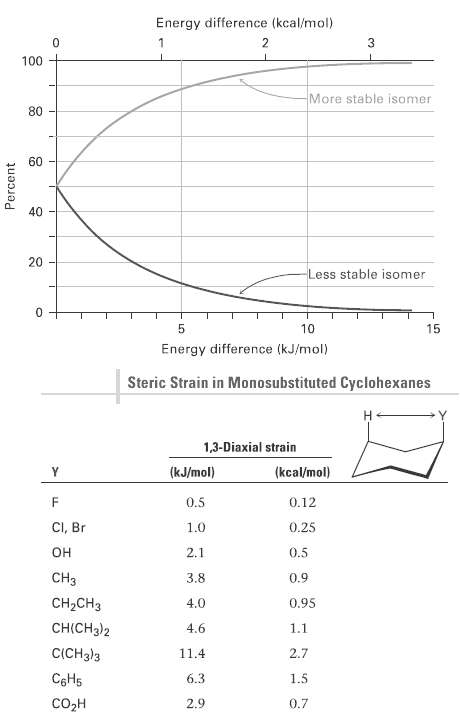
From the data in Figure and Table, estimate the percentages of molecules that have their substituents in
Question:
From the data in Figure and Table, estimate the percentages of molecules that have their substituents in an axial orientation for the following compounds:
(a) Isopropylcyclohexane
(b) Fluorocyclohexane
(c) Cyclohexane carbonitrile,C6H11CN
Transcribed Image Text:
Energy difference (kcal/mol) 2 100 More stable isomer 80 60 40 20 Less stable isomer 10 15 Energy difference (kJ/mol) Steric Strain in Monosubstituted Cyclohexanes Н 1,3-Diaxial strain (kcal/mol) (kJ/mol) 0.5 0.12 CI, Br 0.25 1.0 он 0.5 2.1 CHз 3.8 0.9 CH-CHз 4.0 0.95 CH(CH3)2 4.6 1.1 C(CH3)3 11.4 2.7 C6H5 6.3 1.5 содн 2.9 0.7 Percent
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (14 reviews)
Strategy To solve this problem 1 Find the energy cost of a 13d...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
From the data in Table 15.2, what can you say in general about the ratio of saturated to unsaturated acids in fats and oils?
-
The data in Figure 7.15, Table 7.19, and Table 7.20 are provided for end items A and H. Develop MRP records for items B, C, I, and E. Are there any action notices for the first week? In figure 7.15 ...
-
From the data in Table 14-6, estimate the ratio by which the yield strength of magnesium can be increased by alloying and heat treatment and compare with that of aluminum alloys.
-
With a global economy, the only way to achieve cost-effective control of greenhouse gases is to assure that every country imposes the same universal set of emissions standards. Discuss.
-
According to the financial statements for Samson Electronics, Inc., the firm has total assets valued at $310 million. It also has total liabilities of $190 million. Company records indicate that the...
-
Vending machines reject coins based on weight. Refer to Data Set 21 in Appendix B and use a 0.05 significance level to test the claim that the mean weight of pre-1964 quarters is equal to the mean...
-
Norm Johnson Novelty Company identified the following activities in its pro- duction and support operations. Classify each of these activities as either value-added or nonvalue-added. (a) Purchasing....
-
Quark Model of the Neutron The neutron is a particle with zero charge. Nonetheless, it has a nonzero magnetic moment with z component 9.66 X 10-27 A. m2. This can be explained by the internal...
-
Packaging Solutions Corporation manufactures and sells a wide variety of packaging products. Performance reports are prepared monthly for each department. The planning budget and flexible budget for...
-
Determine whether or not F is a conservative vector field. If it is, find a function f such that F = f. F(x, y) = ye x i + (e x + e y ) j
-
A Draw 1, 3, 5-trimethy1cyclohexane using a hexagon to represent the ring. How many cisTrans stereoisomers are possible?
-
A Assume that you have a variety of cyclohexanes substituted in the positions indicated. Identify the substituents as either axial or equatorial. For example, a 1, 2-cis relationship means that one...
-
Completed-Contract Method) Monat Construction Company, Inc., entered into a firm fixed price contract with Hyatt Clinic on July 1, 2010, to construct a four-story office building. At that time, Monat...
-
The general form is: df+g(t)-f(t)=h(t) dt where g(t) and h(t) are understood to be known. It turns out that any first-order differential equation is relatively easy to solve using computational...
-
Strategic Decision-Making: Innovation and Implementation of a New Reporting Solution Tatjana Vasiljeva, RISEBA University of Business, Arts and Technology, Latvia A beautiful sunny winter day......
-
Reflect on a time you have seen language interfere with a message either at work, school or in your personal life. How might a change in the formality, simplicity, or reduction in redundancy have...
-
A car hydroplaning on wet pavement undergoes deceleration of a(v) = cv, where c = 0.4s 1 , where s is seconds. If the initial velocity of the car is 100 km/hr, determine its displacement as a...
-
Adipoyl chloride reacts violently with water to form adipic acid and HC1. Why does the adipoyl chloride/cyclohexane solution not immediately decompose when it is mixed with an aqueous sodium...
-
True or False: Whenever r is calculated on the basis of a sample, the value which we obtain for r is only an estimate of the true correlation coefficient which we would obtain if we calculated it for...
-
At 31 December 20X9, the end of the annual reporting period, the accounts of Huron Company showed the following: a. Sales revenue for 20X9, $ 2,950,000, of which one- quarter was on credit. b....
-
Calculate the vapor pressure of water at 35.0 C using data from Tables 12.4 and 12.5. Tables 12.4 Tables 12.5 TABLE 12.4 Intermolecular Forces and Enthalpies of Vaporization at 298 Ka Liquid Dimethyl...
-
(a) Draw resonance structures for the carbocation that could be formed from (E)-2-butenyl trifluoromethanesulfonate. (b) One of the resonance structures for this carbocation should be a more...
-
What products would you expect from the reaction of 1 mol of 1,3-butadiene and each of the following reagents? (If no reaction would occur, you should indicate that as well.) (a) 1 mol of Cl2 (b) 2...
-
Provide the reagents necessary to transform 2,3-dimethyl-1,3-butadiene into each of the following compounds. (a) (b) (c) (d) Br
-
Required: Following Simplicity Financial and Business Advisory Group Pty Ltd policies and procedures prepare the following: Task 1 Financial statement analysis The client needs a review of financial...
-
SCC Company reported the following for the current year.Net sales$ 57,000Cost of goods sold47, 200Beginning balance in inventory2,900Ending balance in inventory8,960Compute (a) inventory turnover and...
-
You are an assistant financial consultant for Simplicity Financial and Business Advisory Group PtyLtd, and you have been notified the client converted their business to a public company PTCG Ltd....

Study smarter with the SolutionInn App


