Ultraviolet spectroscopy of often used to monitor the amount of a protein in a sample, the amount
Question:
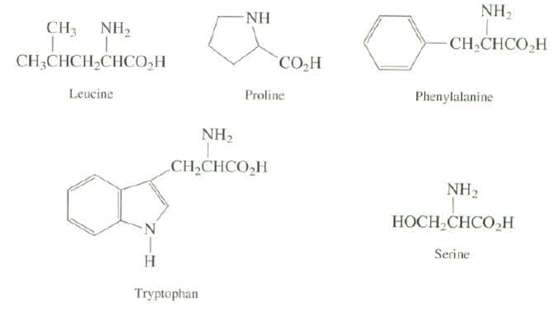
Ultraviolet spectroscopy of often used to monitor the amount of a protein in a sample, the amount of protein is correlated with the absorbance at 280nm. Explain which of the following amino acids you expect to have an absorption at 280nm, (the carbonyl groups of the peptide [amide] bond of the protein do not absorb at thiswavelength).
Transcribed Image Text:
NH, NH NH, CH3 CH CHCO,H CH,CHCH,CHCO,H CO,H Leucine Proline Phenylalanine NH2 CH,CHCO,H NH, НОСН-СHCO,H Serine H. Tryptophan
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 30% (10 reviews)
To have an absorption at 280 nm the amino acid m...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain how FRET could be used to monitor the association of G"' and adenylyl cyclase following activation of the epinephrine receptor.
-
Which of the following amino acids has a nonpolar side chain? NH2 CHSCH2CH2CH COOH NH2 HSCH CH COOH
-
Which of the following amino acids are likely to be linked to only one species of tRNA? What are their anticodons? (a) Phe (b) Leu (c) His
-
Let y be a variable that takes 0 or 1 value. Write an inequality to describe if y = 0, then x can take value from 0 to 5 and if y = 1, then x can take value 0. We are given 3 options. We are allowed...
-
Sales have decreased for two consecutive quarters at your business. You have been instructed to inform your sales team that their hours, and base pay, will be reduced by 20 percent. While you may...
-
Consider an ideal Rankine cycle using water with a high-pressure side of the cycle at a supercritical pressure. Such a cycle has a potential advantage of minimizing local temperature differences...
-
What does Paint by Number Engineering refer to? What are its pros and cons?
-
Orchard Company has monthly salaries of $10,000. Assume Orchard pays all the standard payroll taxes and no employees have reached the payroll tax limits. Journalize the accrual and payment of...
-
You are the CFO at ABC Inc., a public company listed in Toronto Stock Exchange (TSX). Its basic business is high-temperature treatment services for contaminated soil. It is now December 9 th , 2022,...
-
A store maintains data on customers, products and purchase records in three tables: CUSTOMER, PRODUCT, PURCHASE. The store manager wants to know which product is on its maximum discount for each...
-
The 1H-NMR spectrum of compound H is as follows, the peaks at highest m/z in the mass spectrum of H appear at 122 and 124 with intensities in a ration of about 1 to 1. Show the structure ofH. 2 H ...
-
Explain why the ultraviolet spectrum of one of these dienes has its maximum absorption at a longer wavelength than that of the other.
-
Go to Extended Case 28.2, Overseas Private Investment Corp. v. Kim, 69 A.D.3d 1185, 895 N.Y.S.2d 217 (2010) on pages 551552. Read the excerpt and answer the following questions. (a) The main issue...
-
The Holistic Marketing Concept is driving organisations'approaches to marketing in the twenty-first century. The concept recognises the scope and complexities of marketing activities and acknowledges...
-
1. A Chinese company offered to a British counterpart in London at USD500per case FOB Shanghai. The British importer asked the exporter to offer a CIF price. Suppose the freight is USD50 per case and...
-
As a class, discuss the following questions: a. Did the questions you asked provide you with the information you had hoped for? b. Discuss at least one response that you found surprising or...
-
A hollow circular column carries a projecting bracket, which supports a load of 25 kN as shown in Fig. 4.29. The distance between the axis of the column and the load is 500 mm. The inner diameter of...
-
1. Use the cash flow diagram to determine the uniform annual amount in years 1 through 8 that is equivalent to all of the cash flows shown. Use i = 10% per year. [5] -1 0 1 2 3 4 5 6 7 8 Year $25 $25...
-
(a) What percentage of the people live the greatest distance from the store? (b) What is the percentage of people who live 30 or more miles from the store? (c) Once the peak distance of 1 to 5 miles...
-
Coastal Refining Company operates a refinery with a distillation capacity of 12,000 barrels per day. As a new member of Coastal's management team, you have been given the task of developing a...
-
In 2008, the financial markets and banking system of the United States nearly collapsed completely as a result of the issuance and securitization of subprime mortgages. When the housing market began...
-
What is the conjugate acid of each of the following bases? (a) HSO4- (b) H2O (c) CH3NH2 (d) -NH2 (e) CH3CH2 (f) CH3CO2-
-
Designate the Lewis acid and Lewis base in each of the following reactions: (a) (b) (c) CI CH,CH2 CIAIC CH OHBFCH3 CH3 CHs CH
-
Rewrite each of the following reactions using curved arrows and show all nonbonding electron pairs: (a) (b) (c) CH NH2 HC
-
Discuss the innovative idea/project addressing the problem below by the following (a) potential impact - (b) novelty and innovation- (c) feasibility- (d) sustainability- Reference: i. Potential...
-
Marianne's duplex sold at a foreclosure auction for $280,000. At the time of foreclosure, she had not made any payments to the bank for 11 months ($2,400 each). The outstanding balance of her...
-
Find the complete solution of the linear system, or show that it is inconsistent. (If the system has infinitely many solutions, express your answer in terms of t, where x = x(t), y = y(t), and z=t....

Study smarter with the SolutionInn App


