Horizontal analysis (trend analysis) percentages for Algy Company?s sales, cost of goods sold, and expenses are shown
Question:
Horizontal analysis (trend analysis) percentages for Algy Company?s sales, cost of goods sold, and expenses are shown below.
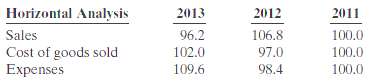
Did Algy?s net income increase, decrease, or remain unchanged over the 3-year period?
Horizontal Analysis 2013 96.2 102.0 109.6 2012 2011 100.0 100.0 Sales Cost of goods sold Expenses 106.8 97.0 98.4 100.0
Step by Step Answer:
Comparing the percentages presented results in the following conclusions T...View the full answer



Accounting Principles
ISBN: 978-0470534793
10th Edition
Authors: Jerry J. Weygandt, Paul D. Kimmel, Donald E. Kieso
Related Video
The Dupont analysis is an expanded return on equity formula, calculated by multiplying the net profit margin by the asset turnover by the equity multiplier. The DuPont analysis is also known as the DuPont identity or DuPont model.This Video will guide on how to calculate return on Equity and estimate profitability of shareholders using DuPont Analysis.
Students also viewed these Managerial Accounting questions
-
Suppose you are buying a car of $26000. The dealer offers that you pay a down payment of $4000 now. Then you will pay the balance with equal monthly payments for 42 months with 6% APR compounded...
-
The Robinson Company had a cost of goods sold of $1,000,000 in 2014 and $1,200,000 in 2015. a. Calculate the inventory turnover for each year. Comment on your findings. b. What would have been the...
-
Cubitene is a diterpene present in the defense secretion of a species of African termite. What unusual feature characterizes the joining of isoprene units in cubitene?
-
Use regression analysis to fit a quadratic trend model to the data set. a. What is the estimated regression function? b. Compare the adjusted-R2 value for this model to that of the linear trend...
-
For liquid water the isothermal compressibility is given by; where r and b are functions of temperature only. If 1 kg of water is compressed isothermally and reversibly from I to 500 bar at 60(C. how...
-
Julie Palmer has trouble keeping her debits and credits equal. During a recent month, Julie made the following errors: a. Julie recorded a $750 payment of rent by debiting rent expense for $75 and...
-
You have been assigned the task of evaluating two mutually exclusive projects with the following projected cash flows: If the appropriate discount rate on these projects is 10 percent, which would be...
-
1.) One year ago, ZXY deposited $1,860 in an account that has earned and will earn 1.90 percent per year in compound interest. If ACB deposits $1,910 in an account today that earns simple interest,...
-
Jones, the investor, proposes taking participating preferred stock1 with 1x liquidation preference in return for her $5 million investment. Draw the payoff diagram for this security from Jones...
-
If Vansittart Company had net income of $585,000 in 2013 and it experienced a 30% increase in net income over 2012, what was its 2012 net income?
-
Selected condensed data taken from a recent balance sheet of Evangelista Inc. are as follows. EVANGELISTA INC. Balance Sheet (partial) Cash..............$ 8,041,000 Short-term...
-
For the data in Problem 14, summarize the total sales of parts and computers. Data from in problem 14 The file named Ptablepartsdata.xlsx contains sales of various parts. Each parts code begins with...
-
How are sales margin variances calculated.
-
Why is majority rule so popular when consensus decision making, by comparison, is often more advantageous?
-
How do communication styles of conflict management differ on task and social dimensions of small groups?
-
Define feed forward control.
-
What are the disadvantages of divisionalisation.
-
What are the three primary methods of cost allocation? How do they differ?
-
Create a data model for one of the processes in the end-of-chapter Exercises for Chapter 4. Explain how you would balance the data model and process model.
-
For the reaction K eq = 3.59 at 900C. After the reaction has run for 10 min at 900 C, the concentrations are [CH 4 ] = 1.15 M; [H 2 S] = 1.20 M; [CS 2 ] = 1.51 M; [H 2 ] = 1.08 M. Is this reaction at...
-
What are the strengths of the annual rate of return approach? What are its weaknesses?
-
What are the strengths of the annual rate of return approach? What are its weaknesses?
-
Your classmate, Mike Dawson, is confused about the factors that are included in the annual rate of return technique. What is the formula for this technique?
-
What are the editorial and design differences between a Web magazine and a print magazine? What in-house marketing groups have to coordinate efforts to plan and execute Ogdens IMC campaigns and...
-
Compare and contrast green marketing vs sustainability marketing using advertisements (TV or print media) of your choice. with 2 references
-
How can a marketing strategy be comprehensive? Does print media have same priority as digital? Explain and give examples of the following ways to promote or impact your product marketing. Brochures...

Study smarter with the SolutionInn App


