How could you use Diels?Alder reactions to prepare the following products? Show the starting diene and dienophile
Question:
How could you use Diels?Alder reactions to prepare the following products? Show the starting diene and dienophile in each case.
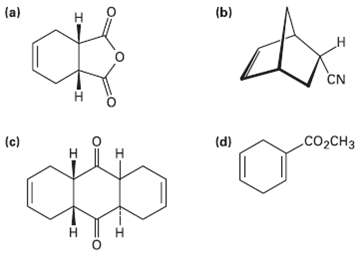
Transcribed Image Text:
(a) (b) н CN сооCнз (c) (d) н H. н т
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
First find the cyclohexene ring formed by the DielsAlder ...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How could you use 1H NMR spectroscopy to distinguish among the following esters? 018 18 - CH3 OH
-
How could you use expectancy theory to increase your own motivation level?
-
How could you use standard process costing to obtain information that helps improve the efficiency of the process?
-
An examiners close inspection of the annual financial statements and the accounting records revealed that Mawani Inc. may have violated some accounting principles. The examiner questioned the...
-
Discuss how well you think Starbuck's tuition reimbursement program meets the criteria for selecting employee benefits (organizational objectives, employees' expectations and values, and benefits...
-
Leno sells tire rims. Its sales budget for the nine months ended September 30 follows: In the past, cost of goods sold has been 60% of total sales. The director of marketing and the financial vice...
-
A plant asset purchased by Krest Inc. for \(\$ 100,000\) late in 2018 is to be depreciated as follows. In 2020 , taxable income was \(\$ 450,000\) and the tax rate is \(25 \%\). Future enacted tax...
-
Presented below is the trial balance of Scott Butler Corporation at December 31, 2014. Instructions Prepare a balance sheet at December 31, 2014, for Scott Butler Corporation. (Ignore incometaxes.)...
-
You are the auditor partner on the engagement. You consider the following independent and material situation: i. Pine Farms Ltd is an online retailer of body lotions. The company records revenue at...
-
Find the transfer function, 1 (s)/T(s) , for the system shown in Figure P2.19. T(1) 0(1) 1 N-m/rad 1 N-m/rad 1 kg-m? 0000 1 kg-m? 1 N-m-s/rad FIGURE P2.19
-
1, 3-Pentadiene is much more reactive in Diels?Alder reactions than 2, 4-pentadienal. Why might this be? H 1,3-Pentadiene 2,4-Pentadienal
-
Aldrin, a chlorinated insecticide now banned for use in the United States, can be made by Diels?Alder reaction of hexachloro-1, 3-cyclopentadiene with norbornadiene. What is the structure of aldrin?...
-
Bishop, Inc., has current assets of $5,700, net fixed assets of $27,000, current liabilities of $4,400, and long-term debt of $12,900. What is the value of the shareholders equity account for this...
-
For this messaging strategy practice exercise, you'll assume the role of a marketing consultant hired by the Centers for Disease Control and Prevention (CDC). Your job is to design a public health...
-
Poor Mr.Wang He has been struggling with his injuries. The skin on his hands degenerated and required skin grafts. The skin graft on his dominant hand got a staph infection. After 8 months of...
-
Determining Friction from the Stopping Distance 1. The simulation The code below shows what happens to a system that has an initial velocity, and then slides to a stop due to friction. 1. Try...
-
Can you determine whether an investment is warranted based solely upon the NPV? If three mutually exclusive investments all show about the same NPV, how might you determine which to pursue?
-
e. Identify the highest favorable variance and highest unfavorable variance from the six listed in requirement d, and provide one possible cause of each variance.
-
A unidirectional IM-9 carbon fiber/Hexply 8551-7 epoxy composite is to be designed to replace a 6061-T6 aluminum alloy rod which is to be loaded in longitudinal tension. (a) What fiber volume...
-
(a) Prove that form an orthonormal basis for R3 for the usual dot product. (b) Find the coordinates of v = (1, 1, 1)T relative to this basis. (c) Verify formula (5.5) in this particular case. 48-65...
-
A solution contains a mixture of substance A and substance B, both of which are volatile. The mole fraction of substance A is 0.35. At 32 C the vapor pressure of pure A is 87 mmHg, and the vapor...
-
Propose a mechanism for the acid-catalyzed reaction of benzaldehyde with methanol to give benzaldehyde dimethyl acetal.
-
Propose a mechanism for the acid-catalyzed hydrolysis of cyclohexanone dimethyl acetal.
-
Why were no products from McLafferty rearrangement observed in the spectrum of butan-2-one (Figure 18-3)? In Figure 18.3 100 43 80 0 60 CH-C CH2CH 40 20 57 0 10 20 30 40 50 60 70 80 90 100 110 120...
-
subject to: Maximize 6x + 14x2 + 13x3, x+2x2+4x324 (Metalworking capacity), x1+2x2+4x360 (Woodworking capacity), x10, x2 0, x3 0, obtaining an optimal tableau:
-
8. Technician A says that MIG stands for metal ignit- able gas. Technician B says that gas metal arc welding (GMAW) is a more accurate term than MIG. Who is correct? A. Technician A only B....
-
When services are performed on? account: A. cash is increased. B. accounts payable is increased. C. accounts receivable is increased. D. revenue will not be recorded until the cash is received from...

Study smarter with the SolutionInn App


