How would you carry out the following transformations? Co CH2 C Lc
Question:
How would you carry out the following transformations?
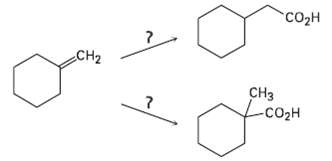
Transcribed Image Text:
"Coон CH2 CНз Lcозн
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
a CH H CH2OH CHBr PBr3 1 BH3 THF 2 HO OH Grignard carboxylation ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you carry out the following transformations using an organo copper coupling reaction? More than one step is required in eachcase. (a) "CH (b) HH2CH2CHBr CH3CH2CH2CH2CH2CH2CH2CH3 (c)...
-
How would you carry out the following transformations? More than one step may berequired. H-CH (a) (b) Br - (c) " - " - (d) CH3CH2CH2CH2C=CH CHH2CH2CH2CH2H20CH3 H (e) CH3CH2CH2CH2CHCH3...
-
How would you carry out the following transformation, a step used in the commercial synthesis of(S)-ibuprofen? CN
-
Which do you feel is more important for a new business: the income statement or the statement of ash flow? Why?
-
Ask an experienced leader what he or she thinks is the most effective method of developing leadership skills. Bring your findings back to class.
-
Use the relative ionic radii in Fig. 13.8 to predict the structures expected for CsBr and KF. Do these predictions agree with observed structures? The ionic radius of Cs+ is 169 pm. Figure 13.8 Lit...
-
How can the lifecycle approach be used to maximise return from a product?
-
Moriarity and Holmes enter into an oral contract by which Moriarity promises to sell and Holmes promises to buy Blackacre for $100,000. Moriarity repudiates the contract by writing a letter to Holmes...
-
2 2 points eBook Hint Ask Print Exercise 13-12 (Static) Analyzing profitability LO P3 Simon Company's year-end balance sheets follow. At December 31 Assets Cash Accounts receivable, net Merchandise...
-
Determine taxable income in each of the following independent cases. In all cases, the company was very profitable in all years prior to 2017 and it had retained earnings of $1,000,000 at the end of...
-
Using 13 CO 2 as your only source of labeled carbon, along with any other compounds needed, how would you synthesize the following compounds? (a) CH 3 CH 2 13 CO 2 H (b) CH 3 13 CH 2 CO 2 H
-
Which method?Grignard carboxylation or nitrile hydrolysis?would you use for each of the following reactions? Explain. " - C CH2Br (a) " CH Br (b) CH3CH2CHCO2H CCH2CHCH3 (c) CHH-H2H2, CHH-H-CH21...
-
Describe three standard therapies for cancer treatment?
-
Question 3 ESG Limited ("ESG") manufactures electric motors for various brands of machines. The motors are designed and assembled according to customers' specifications. ESG uses normal costing to...
-
Benoit Company produces three products-A, B, and C. Data concerning the three products follow (per unit): Product B Selling price Variable expenses: Direct materials Other variable expenses Total...
-
Jackson Autos has one employee. As of March 30, their employee had already earned $6,120. For the pay period ending April 15, their employee earned an additional $2,040 of gross wages. Only the first...
-
Laura currently manages the polished chrome division of Blue Broadway, a business that specializes in ceiling light fixtures. Its performance has been stable for the past few years. However, the...
-
Use the following consolidated balance sheet for Campbell Soup Company annual report. CAMPBELL SOUP COMPANY Consolidated Balance Sheets (Millions, except per share amounts) Current assets July 30,...
-
Is this a right-tailed, left-tailed, or two-tailed test? The U.S. Center for Disease Control reports that the mean life expectancy was 47.6 years for whites born in 1900 and 33.0 years for nonwhites....
-
Complete the following acid-base reactions: (a) HCCH + NaH
-
Classify each of the following as a component of a silicate ceramic, an oxide ceramic, or a nonoxide ceramic. a. B 4 C b. Mg 2 SiO 4 c. MoSi 2
-
The guanidino group of arginine is one of the most strongly basic of all organic groups. Explain. NH NI NI
-
(a) dl-Glutamic acid has been synthesized from diethyl acetamidomalonate in the following way. Outline the reactions involved. (b) Compound G has also been used to prepare the amino acid dl-ornithine...
-
Synthetic polyglutamic acid exists as an a helix in solution at pH 2-3. When the pH of such a solution is gradually raised through the addition of a base, a dramatic change in optical rotation takes...
-
Way Cool produces two different models of air conditioners. The company produces the mechanical systems in its components department. The mechanical systems are combined with the housing assembly in...
-
Hatch Manufacturing produces multiple machine parts. The theoretical cycle time for one of its products is 30 minutes per unit. The budgeted conversion costs for the manufacturing cell dedicated to...
-
The objective of the Additional Refundable Tax on Investment Income is to discourage the use of a Canadian controlled private corporation to defer taxes on investment income. . True False

Study smarter with the SolutionInn App


