How would you prepare the following substances from 2-cyclohexcnone? More than one step may be required. .CH
Question:
How would you prepare the following substances from 2-cyclohexcnone? More than one step may be required.
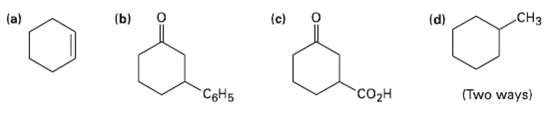
Transcribed Image Text:
.CHз (a) (b) (d) (c) сон CSH5 (Two ways)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
do HNNH KOH b d 1 LiC6H52 Cu ...View the full answer

Answered By

David Muchemi
I am a professional academic writer with considerable experience in writing business and economic related papers. I have been writing for my clients who reach out to me personally after being recommended to me by satisfied clients.
I have the English language prowess, no grammatical and spelling errors can be found in my work. I double-check for such mistakes before submitting my papers.
I deliver finished work within the stipulated time and without fail. I am a good researcher on any topic especially those perceived to be tough.
I am ready to work on your papers and ensure you receive the highest quality you are looking for. Please hire me to offer my readily available quality service.
Best regards,
4.60+
27+ Reviews
61+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you prepare the following substances from Cyclopentanol? More than one step may be required. (a) Cyclopentanone (b) Cyclopentane (c) 1-Methylcyclopentanol (d) Trans-2-Methylcyclopentanol I...
-
How would you prepare the following substances from 1-butanol? (a) Butyl amine (b) Dibutyl amine (c) Propyl amine (d) Pentylamine (e) N, N-Dimethyl butyl amine (f) Propene
-
How would you prepare the following substances from Pentanoic acid? (a) Pentanamide (b) Butyl amine (c) Pentylamine (d) 2-Bromopentanoic acid (e) Hexane nitrile (f) Hexylamine
-
Sherene Nili manages a company that produces wedding gowns. She produces both a custom product that is made to order and a standard product that is sold in bridal salons. Her accountant prepared the...
-
1. Review the first section of the chapter, about becoming world class. How can EatWell Technologies fulfill its social mission and be profitable? 2. In this case, where do you see resistance to...
-
The electron affinities of the elements from aluminum to chlorine are 244 kJ/ mol, 2120 kJ/ mol, 274 kJ/ mol, 2200.4 kJ/ mol, and 2348.7 kJ/ mol, respectively. Rationalize the trend in these values.
-
In Cleo Company, the predetermined overhead rate is 80% of direct labor cost. During the month, \($210,000\) of factory labor costs are in- curred, oi which \($180,000\) is direct labor...
-
Bert founded Sambert Corporation a little over a year ago. He believes that his company, which sells specialized computer toys, will be very profitable over the next several years, as evidenced by...
-
ch Question 2 60 pts For each of the following transactions, indicate the appropriate section of the statement of cash flows. A. Operating Section B. Investing Section C. Financing Section D....
-
On January 1, 2020, Daso Ltd. entered into an agreement to lease a truck from Henry Ltd. Both Daso and Henry use IFRS 16. The details of the agreement are as follows: Carrying value of truck for...
-
Predict the products of the reaction of (i) phenyl acetaldehyde and (ii) acetophenone with the following reagents: (a) NaBH4 then H3O+ (b) Tollens reagent (c) NH2OH, HC1 catalyst (d) CH3MgBr, then...
-
Show how the Wittig reaction might be used to prepare the following alkenes. Identify the alkyl halide and the carbonyl components that would he used. (b) (a)
-
Comment on the following field situations and make recommendations for corrective action. a. One of the interviewers has an excessive rate of refusals in in-home personal interviewing. b. In a CATI...
-
The brakes are applied to a moving vehicle, causing it to uniformly slow down. While slowing, it moves a distance of 4 0 . 0 m in 7 . 8 0 s to a final velocity of 1 . 3 5 m / s , at which point the...
-
(Marginal Cost of Capital (MCC), Investment Opportunity Schedule (IOS) and Financing Break points (BPs) Caldwell Products Limited (CPL) has compiled the following data for its three sources of...
-
Use your market research to draft evaluation criteria for the acquisition. Keep the following aspects of a potential vendor proposal in mind as a starting point: quality (e.g., experience and...
-
Oslo Company prepared the following contribution format income statement based on a sales volume of 1,000 units (the relevant range of production is 500 units to 1,500 units): Sales Variable expenses...
-
Prepare journal entries for each of the transactions. ( Credit account titles are automatically indented when amount is entered. Do not indent manually. Record journal entries in the order presented...
-
Is the traveler disputing the claim about the average or about the variance? Suppose an airline claims that its flights are consistently on time with an average delay of at most 15 minutes. It claims...
-
On October 1, 2021, Adoll Company acquired 2,600 shares of its $1 par value stock for $38 per share and held these shares in treasury. On March 1, 2023, Adoll resold all the treasury shares for $34...
-
Which of the following ionic compounds is composed of only nonmetal atoms? (a) NH 4 NO 3 ; (b) Al 2 (SO 4 ) 3 ; (c) Na 2 SO 3 ; (d) AlCl 3 ; (e) None of these.
-
When excess formaldehyde in basic solution is treated with ethanal, the following reaction takes place: Write a mechanism that accounts for the formation of the product.
-
When pseudoionone is treated with BF3 in acetic acid, ring closure takes place and α-and β-ionone are produced. This is the next step in the vitamin A synthesis. (a) Write...
-
(a) Write resonance structures for the anion of acetonitrile that account for its being much more acidic than ethane. (b) Give a step-by-step mechanism for the condensation of benzaldehyde with...
-
4. Qu tipo de empresas podran emplear un mercado de empresas privadas? Qu tipo de empresas podra participar en el mercado de una empresa privada? Cmo es un mercado de empresas privadas diferente de...
-
Camby Corp. can make one of the following four products: Product 1 Product 2 Product 3 Product 4 Selling price per unit $109.00 $186.00 $306.00 $45.00 Variable cost per unit $38.00 $120.00 $191.00...
-
If the order quantity (Q) for raw materials is increased, how does it affect ordering cost and/or holding cost? Group of answer choices Holding cost decreases No impact on ordering and holding costs...

Study smarter with the SolutionInn App


