Identify the chirality centers in the following molecules (yellow-green = Cl, pale yellow =F): (b) (a) Threose
Question:
Identify the chirality centers in the following molecules (yellow-green = Cl, pale yellow =F):
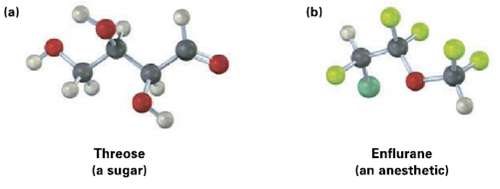
Transcribed Image Text:
(b) (a) Threose (a sugar) Enflurane (an anesthetic)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
a H H H ...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify the chirality centers in the following molecules. Build molecular models if you needhelp. (b) (c) CH30. CH2CH2CH3 C (a) - Coniine (poison hemlock) N-CH3 Menthol (flavoring agent)...
-
Assign R or S stereochemistry to the chirality centers in the following Newmanprojections: CI (a) (b) C "CH . "
-
Assign R or S configurations to the chirality centers in the followingmolecules: CI H la) (c) (b) H
-
Outline suggestions to make observation a useful, reliable requirements elicitation technique.
-
Discuss why sound travels faster in moist air. (Note that at the same temperature, water vapor molecules have the same average kinetic energy as the heavier nitrogen and oxygen molecules in the air.)...
-
What is the overhead flexible-budget variance?
-
Which type of adjustment will (a) increase both assets and revenues, (b) increase revenues and decrease liabilities, (c) increase expenses and decrease assets, and (d) increase both expenses and...
-
A firm wishing to evaluate interest rate behavior has gathered data on the nominal rate of interest and on inflationary expectations for five U.S. Treasury securities, each having a different...
-
Use the given information and your calculated numbers to complete the Cost of Goods Sold Equation below for all three inventory methods. ( All numbers should be positive. ) ( Use cells A 4 to D 1 0...
-
The room temperature of an 11 m 2 room is to be controlled by varying the power of an indoor radiator. For this specific room, the open-loop transfer function from radiator power, Q (s), to...
-
Alanine, an amino add found in proteins, is chiral. Draw the two enantiorners of ala- nine using the standard convention of solid, wedged, and dashedlines. NH2 Alanine CHO2H
-
Is cocaine (Worked Example 9.2) dextrorotatory or levorotatory?
-
Rapidly growing strong economies often experience trade deficits, whereas economies with sluggish growth often have trade surpluses. Can you explain this puzzle?
-
Describe how payors utilize codes such as DRGs and ICD10s to support claims/billing activities? What are the implications of improper coding
-
As a local government department head, how knowledgeable should you be in the various financing options available for purchasing capital equipment? Explain your reasoning.
-
In 2023, Lo sold his chalet on Lac des Deux Montagnes for $134,000 in cash. He acquired this chalet in 2013 for $79,000. Lo spent at least one month a year in this chalet. He wonders if this cottage...
-
1. The current par yield (the yield y that solves ParValue = +++ + 1+y (1+y) c is the coupon payment at the end of each year) curve is given by: C ParValue+c + + where (1+y)n Maturity (in year) 1 2 3...
-
Consider the following and show all work. Point Line (4, -9) 2x-3y = -6 a) Write the slope-intercept form of the equation of the line through the given point and parallel to the given line. (b) Write...
-
Before acquiring Unicru, Kronos had specialized in other segments of the human resources technology field, such as time-and-attendance applications and payroll systems.127 Its other products are...
-
What are some of the features of the Unified Process (UP)?
-
Methanol (CH 3 OH) can be synthesized by the reaction: What volume (in liters) of hydrogen gas, at a temperature of 355 K and a pressure of 738 mmHg, is needed to synthesize 35.7 g of methanol? CO(g)...
-
Predict the product when each of the following compounds reacts with one equivalent of lithium dimethylcuprate, followed by protonolysis. Explain. CH,(CH,),0-C(CH,),C-CI
-
Outline two methods for the preparation of 5-methylhexanoic acid from 1-bromo-4-methylpentane.
-
One interesting process for making nylon-6,6 demonstrates the potential of using biomass as an industrial starting material. The raw material for this process, outlined in the following reaction, is...
-
Cost-Volume-Profit Analysis at General Electric Values: Item Amount Selling Price $100,000 Variable Cost $80,000 Fixed Costs $15,000,000 Requirements: Calculate the break-even point in units....
-
Analyze the cost-volume-profit relationship for Coca-Cola. The company sells its beverage product for $2 per unit. The variable cost per unit is $1.20, and the total fixed costs are $50,000....
-
"This is really an odd situation," said Jim Carter, general manager of Highland Publishing Company. " We get most of the jobs we bid on that require a lot of press time in the Printing Department,...

Study smarter with the SolutionInn App


