Identify the indicated faces of carbon atoms in the following molecules as Re orSi: (b) (a) H-2
Question:
Identify the indicated faces of carbon atoms in the following molecules as Re orSi:
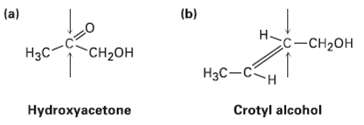
Transcribed Image Text:
(b) (a) Hс-сн2он "CH2он Нас- Hас— Сн Hydroxyacetone Crotyl alcohol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
Strategy Draw the plane that includes the sp carbon and its substituents and ...View the full answer

Answered By

Aun Ali
I am an Associate Member of Cost and Management Accountants of Pakistan with vast experience in the field of accounting and finance, including more than 17 years of teaching experience at university level. I have been teaching at both undergraduate and post graduate levels. My area of specialization is cost and management accounting but I have taught various subjects related to accounting and finance.
5.00+
13+ Reviews
32+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify the carbon atoms in the following molecules as primary, secondary, tertiary, orquaternary: H H CH CH CHCH2CHCH2CH Gs (c) (a) (b) CH2H2CH CHCCH2CCH CH
-
Identify the indicated faces in the following molecules as Re orSi:
-
Identify the indicated hydrogen?s in the following molecules as pro-R or pro-S: (a) (b) .co2 H3N Alanine (S)-Glyceraldehyde
-
Listed below are selected items from the cost-of-quality (COQ) report for Watson Products for last month. Category Amount Rework ........... $ 725 Equipment maintenance ...... 1,154 Product testing...
-
Wrap a piece of paper around a thick metal bar and place it in a flame. Notice that the paper will not catch fire. Can you explain this in terms of the conductivity of the metal bar? (Paper generally...
-
A sample of 100 randomly selected registered voters in the city of Naperville was asked their political afï¬liation: Democrat (D), Republican (R), or Independent (I). The results of the...
-
Question: Hank rents an apartment in a rental housing community called Mountain Garden, a middle-class development of five buildings. Crime on the estate is about 15 percent lower than in the...
-
A partial trial balance of Julie Hartsack Corporation is as follows on December 31, 2015. Additional adjusting data: 1. A physical count of supplies on hand on December 31, 2015, totaled $1,100. 2....
-
I am trying to practice creating Statement of cashflows.. I made an attempt and would like to ask for a tutor to see if my attempt is correct (advise of the things I might have missed) and would like...
-
KCCO, Inc., has current assets of $5,300, net BASIC fixed assets of $24,900, current liabilities of $4,600, and long-term debt of $10,300. What is the value of the shareholders' equity account for...
-
What products are formed from hydration of 4-methylcyclopentene? What can you say about the relative amounts of the products?
-
Lactic acid buildup in tired muscles results from reduction of pyruvate. If the reaction occurs from the Re face, what is the stereochemistry of theproduct? OH CH3CHCO2 H3C CO2 Lactate Pyruvate
-
The total drag coefficient for an airplane wing is C D = C D0 + C 2 L /, where CD0 is the form drag coefficient, C L is the lift coefficient and is the aspect ratio of the wing. The power is given...
-
A company has $20 million face value of bonds outstanding that pay a coupon of 10 percent annually and have 8 years to maturity. The bonds have a 12 percent yield to maturity. What value should be...
-
Add. State the sum in simplest form. 8 12/13 + 19/34 2x 9y
-
Discuss the processes and structures that are important to the organization's goals and objectives or job role. 2. Why should you develop procedures and structures that are connected to the...
-
Discuss the internal and external forces for change today. Explain how managers can serve as change agents and describe what organizationAl elements are within a manager's control Explain why the...
-
Wheeling Manufacturing orders 8,000 units of graphite shafts for its production of golf clubs per week. The carrying costs of these shafts are $5 per unit per year and the fixed ordering cost is...
-
Using a computer simulation, verify the results of Example 4.7 for the source-follower amplifier. Data From Example 4.7:- Calculate the small-signal voltage gain of the source-follower circuit in...
-
KD Insurance Company specializes in term life insurance contracts. Cash collection experience shows that 20 percent of billed premiums are collected in the month before they are due, 60 percent are...
-
Calculate the number of moles of gas in a 3.24-L basketball inflated to a total pressure of 24.3 psi at 25 C. The total pressure is not the same as the pressure read on a pressure gauge such as the...
-
When compound A in Fig. P22.64 is treated with NaOCH3 in CH3OH, isomerization to compound B occurs. Explain why, when compound C is subjected to the same conditions, no isomerization occurs. NaoCHCH...
-
Tropone reacts with one equivalent of HBr to give a stable crystalline conjugate acid salt with a pKa of -0.6, which is considerably greater (that is, less negative) than the pKa values of most...
-
Tropone reacts with one equivalent of HBr to give a stable crystalline conjugate acid salt with a pKa of -0.6, which is considerably greater (that is, less negative) than the pKa values of most...
-
Select the activity below that is not appropriate for the post-reading, "recite and review" stage of SQ3R. Preview next week's textbook reading by looking at the table of contents to predict the...
-
Maple Leaf Inc. uses a periodic inventory system. It entered into the following purchases and sales transactions for May. Date Activities Units Acquired at Cost Units Sold at Retail May 1 Beginning...
-
OSHA must investigate an occupational accident: O If it involves an injury If someone is killed To levy a fine To issue citations

Study smarter with the SolutionInn App


