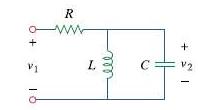
It is desired to realize the transfer function using the circuit in Fig. 16.76. Choose R =
Question:
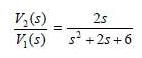
using the circuit in Fig. 16.76. Choose R = 1 kΩ and find L and C.
Transcribed Image Text:
巧(s) V(s) 2s +6 2s
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
Comparing this with the given transf...View the full answer

Answered By

Sumit kumar
Education details:
QUATERNARY Pursuing M.Tech.(2017-2019) in Electronics and Communication Engg. (VLSI DESIGN) from
GNIOT Greater Noida
TERTIARY B.Tech. (2012-2016) in Electronics and Communication Engg. from GLBITM Greater Noida
SECONDARY Senior Secondary School Examination (Class XII) in 2012 from R.S.S.Inter College, Noida
ELEMENTARY Secondary School Examination (Class X) in 2010 from New R.J.C. Public School ,Noida
CERTIFICATION
Summer Training in ‘WIRELESS EMBEDDED SYSTEM’ from ‘XIONEE’ for the six weeks.
EMBEDDED SYSTEM Certificate issued by CETPA INFOTECH for one day workshop.
Certificate of Faculty development program on OPTICAL COMMUNICATION and NETWORKS for one week.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Electric Circuits
ISBN: 9780073301150
3rd Edition
Authors: Matthew Sadiku, Charles Alexander
Question Posted:
Students also viewed these Electricity and Magnetism questions
-
Realize the transfer function V0(s)/Vs(s) = s/s + 10 using the circuit in Fig. 16.78. Let Y1 = sC1, Y2 = 1/R1, Y3 = sC2. Choose R1 = 1k Ω and determine C1 and C2. Y.
-
It is desired to recover hexane vapor (solute A) from air using an absorption process. The absorption solvent is a nonvolatile mineral oil, which has a mass density of 0.80 g/cm 3 and a molecular...
-
An aeration basin is filled to a volume of 425 m 3 with wastewater containing dissolved hydrogen sulfide (H 2 S) of initial concentration 0.300 gmole/m 3 . The aeration basin is equipped with 10 gas...
-
The following data were taken from the year- end records of Glare Import Company: Required: Fill in all of the missing amounts. Show computations. Year 2 Statement of Eamings Items Gross sales venue...
-
A part is loaded with a combination of bending, axial, and torsion such that the following stresses are created at a particular location: Bending: ..Completely reversed, with a maximum stress of 60...
-
What purpose(s) might be served by using each of the following priority rules for allocating scarce resources? a. As late as possible b. Shortest task duration time first c. Minimum slack first
-
Derive Eq. (14-15a) and the corresponding equation for \(\mathrm{x}_{\mathrm{D}, \mathrm{M}}\). (14--15a) XA.M EOYA,0 + FXA,N+1 EO+F 0+(1000) (0.2) 662+1000 = 0.120
-
If the percentage of fuel in a fuelair mixture falls below a certain value called the lower flammability limit (LFL), the mixture cannot be ignited. For example, the LFL of propane in air is 2.05...
-
if you help me can you show me your working steps. Required information Exercise 10-15A (Algo) Straight-line amortization of a bond premium LO 10-5 (The following information applies to the questions...
-
P6-104 Compute gross profit rate and inventory loss using gross profit method Suzuki Company lost all of its inventory in a fire on December 26, 2015. The accounting records showed the following...
-
Determine whether the op amp circuit in Fig. 16.75 is stable. +)
-
Design an op amp circuit, using Fig. 16.77, that will realize the following transfer function: Choose C1 = 10F; determine R1, R2, and C2 (s) s+1000 (s) 2(s+4,000) C2 Ci R2
-
Justify the rule for division of rational numbers.
-
3. A company is trying to optimize its inventory cost. Its conditions are: Annual Demand = 1,500 dozens/year, Ordering cost = $35/order, Holding cost = 40% The supplier is offering the following...
-
A 1 0 - year - to - maturity callable bond paying 6 % interest semi - annually is currently trading at $ 1 , 1 0 0 . The call provision allows the firm to call the bond at the price of $ 1 , 0 4 0 at...
-
Nicholas Jay, Kamla Paul, and Stephanie Ram plan to liquidate their partnership. They have always shared losses and gains in a 1:4:5 ratio, and on the day of the liquidation their balance sheet...
-
Sam would like to make make some plans for his future he is just 25 in graduated and landed a job with a bright future he plans to work for 40 years and once you retire at age 65 based on his salary...
-
Question 1 Faiz was employed by MTD Holdings as a legal officer in 2015 before he was promoted to Senior Legal officer in 2017. In 2019 he was again promoted to be the General Manager of Bayu TD Sdn...
-
MK Manufacturing produces compressor and turbine blades for jet engines. The blades are manufactured from an alloy that is a mix of aluminum and titanium. Sheaths are part of the assembly that keep...
-
Walker, Inc., is an all-equity firm. The cost of the company's equity is currently 11.4 percent and the risk-free.rate is 3.3 percent. The company is currently considering a project that will cost...
-
Write each vector in Fig. 1.34 in terms of the unit vectors i and j. B (15.0 n) 120 30.0 (100m) 53.0 25.0 (12.0m) A (8.00 in)
-
In each case, find the x and y-components of vector 1 : (a) A = 5.Oi - 63j; (b) A = 11.2j - 9.91i; (c) A = -15.Oi +22.4j; (d) A = 5.0B, where B = 4i - 6j.
-
(a) Write each vector in Fig. 1.37 in terms of the unit vectors I and j. (b) Use unit vectors to express the vector C, where C = 3.0M - 4.00B. (c) Find the magnitude and direction of C. A (3.60 m)...
-
Baker Industries' net income is $27,000, its interest expense is $4,000, and its tax rate is 25%. Its notes payable equals $25,000, long-term debt equals $70,000, and common equity equals $250,000....
-
If the interest rate ( SAIR ) is 1 0 % per annum a , What is the effective annual interest rate ( EAIR ) if the compounding frequency is semi - annual? % b , What about if the compounding frequency...
-
1 Problem 9.1 Check my work .28 pints eBook Ask Print Suppose that you hold a piece of land in the city of London that you may want to sell in one year. As a U.S. resident, you are concerned with the...

Study smarter with the SolutionInn App


