Ketones react with acetylide ion (Section 8.7) to give alcohols. For example, the reaction of sodium acetylide
Question:
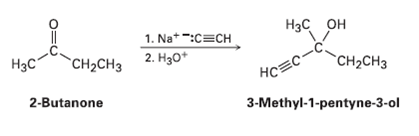
Ketones react with acetylide ion (Section 8.7) to give alcohols. For example, the reaction of sodium acetylide with 2-hutanone yields 3-methyl-l-pentyn-3-ol:
(a) Is the product chiral? Is it optically active?
(b) How many stereo isomers of the product are formed, what are their stereo- chemical relationships, and what are their relativeamounts?
Transcribed Image Text:
1. Na*-:C=CH 2. Hзо* Нзс он Нас "CH-CHз "CH-CHз НоЕс- 2-Butanone 3-Methyl-1-pentyne-3-ol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
The reaction proceeds by addition of acetylide anion to the carbonyl g...View the full answer

Answered By

Amos Kiprotich
I am a wild researcher and I guarantee you a well written paper that is plagiarism free. I am a good time manager and hence you are assured that your paper will always be delivered a head of time. My services are cheap and the prices include a series of revisions, free referencing and formatting.
4.90+
15+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How many stereo isomers of 2, 4-dibromo-3-chloropentane are there? Draw them, and indicate which are optically active.
-
How many stereo isomers exist for this compound? Assign the relative stabilities of each. Is the methyl group axial or equatorial in the more stable conformer of the least stable stereo isomer? CH3 Ph
-
Reaction of lithium diphenylcuprate with optically active 2-bromobutane yields 2 phenyl butane, with high net inversion of configuration. When the 2-bromobutane used has the stereostructure shown,...
-
Lang Enterprises was started when it acquired $4,000 cash from creditors and $6,000 from owners. The company immediately purchased land that cost $9,000. Required a. Record the events under an...
-
Poke a hole in a piece of cardboard and hold the cardboard horizontally in the sunlight (as in Figure 1.6). Note the image of the Sun that is cast below. To convince yourself that the round spot of...
-
Explain the three types of management compensation.
-
Transaction Analysis} Stanfield Inc. entered into the following transactions. a. Issued common shares to investors in exchange for \(\$ 50,000\) cash b. Borrowed \(\$ 15,000\) cash from Royal Bank c....
-
1. Pat Corporation paid $100,000 cash for the net assets of Sag Company, which consisted of the following: Assume Sag Company is dissolved. The plant and equipment acquired in this business...
-
2 . According to the accounting principle of consistency, how should a company treat changes in accounting policies? A . Disclose them in the financial statements B . Implement them without...
-
You are taking a nap by the lake outside. Suddenly, you are startled awake by a strange sound. You swing your head toward the thumping of ducks dancing to The Shake n' Bake. You can't take your eyes...
-
Well see in the next chapter that alkyl halides react with nucleophiles to give substitution products by a mechanism that involves inversion of stereochemistry at carbon: Draw the reaction of...
-
Imagine that another reaction similar to that in Problem 9.81 is carried out between sodium acetylide and (R)-2-phenylpropanal to yield 1-phenyl- 3-butyn-2-ol: (a) Is the product chiral? Is it...
-
Find L-1. S+7] s+2s +5
-
6) f(x) = x - 4 Evaluate lim f(x). x 2 7) Joey the mountain climber is hiking up a mountain with slope 2/3. Using his altimeter he finds that he is gaining altitude at a rate of 6000 feet/hour. How...
-
What is a question that should be asked about accounts payable when forecasting? What is the cash conversion cycle? How much product on hand is too much? How quickly can we replenish goods? How will...
-
Disney has assets worth $250.378 billion. It has debt with a total face value of $38.057 billion. If the risk-free interest rate is 0.375% per annum and the standard deviation of monthly returns on...
-
The price of a telescope purchased by Future Labs Inc. was $32,100. It cost $898 for delivery. The salvage value at the end of 3 years is $6,764. Using the 150% declining-balance depreciation method,...
-
Please explain how income is treated differently in terms of filing taxes than from an accounting perspective?
-
Estimate the modified internal rate of return for the project described in problem 8. Does it change your decision on accepting this project?
-
Respond to the ethical judgments required based on the following scenarios. Scenario 1. Assume you have collected a sample using MUS and that you have evaluated that sample to calculate a total...
-
What is the oxidation state of Cr in each compound? a. Cro b. CrO3 c. CrO3
-
In paper electrophoresis, amino acids and peptides can be separated by their differential migration in an electric field. To the center of a strip of paper is applied a mixture of the following three...
-
Suppose a mixture of AQC-amino acids is subjected to HPLC on a stationary phase that consists of C8-silica rather than Cl 8-silica; that is, the glass stationary phase (Eq. 26.34, p. 1293) contains...
-
Suppose a mixture of AQC-amino acids is subjected to HPLC on a stationary phase that consists of C8-silica rather than Cl 8-silica; that is, the glass stationary phase (Eq. 26.34, p. 1293) contains...
-
Natalie was recruited as the Chief Marketing Officer (CMO) of Beta Corporation, a privately-held company in the consumer goods sector, effective July 1, 2022. Throughout the fiscal year 2023, Beta...
-
A manufacturing company incurred direct materials costs of $50,000, direct labor costs of $45,000, and factory overhead costs of $35,000 during the period. Calculate the total manufacturing costs and...
-
Compute Return on Assets (ROA): Requirements: Using data from a balance sheet and income statement, calculate the return on assets (ROA) with the following figures: Net Income: $100,000 Average Total...

Study smarter with the SolutionInn App


