Ketones undergo a reduction when treated with sodium borohydride, NaBH 4 . What is the structure of
Question:
Ketones undergo a reduction when treated with sodium borohydride, NaBH4. What is the structure of the compound produced by reaction of 2-butanone with NaBH4 if it has an IR absorption at 3400 cm?1 arid M+ = 74 in the mass spectrum?
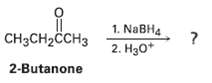
Transcribed Image Text:
1. NABH4 2. Hзо* |CH3CH2CCHЗ 2-Butanone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (15 reviews)
CH3CH2CCH3 1 NaBH4 2 H3O OH ...View the full answer

Answered By

David Ngaruiya
i am a smart worker who concentrates on the content according to my clients' specifications and requirements.
4.50+
7+ Reviews
19+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What is the structure of ATCase?
-
What is the structure of the global beer industry?
-
What is the structure of the beer industry?
-
What do you think people would say about Corrie from the few quotes we have from her book? What was her personality like? Do you think she handled her incarceration differently than Elie Wiesel?...
-
Industries with high barriers to entry often have high barriers to exit. Explain.
-
A regression analysis is carried out, and a confidence interval for 1 is computed to be [1.25, 1.55]; a confidence interval for 2 is [2.01, 2.12]. Both are 95% confidence intervals. Explain the...
-
The Sargent Metal Fabricators has been manufacturing machine tools for a number of years and has an industrywide reputation for high-quality work. The company has been faced with irregularity of...
-
Suppose that peoples expectations of inflation are subject to random shocks. That is, instead of being merely adaptive, expected inflation in period t, as seen in period t 1, is Et1 t = t1 + t1,...
-
Type answer rounded to 1 d.p., with one space between the number and the units. For example, 8.8 fps, 19.2 fps. Help: Lecture 84, Ex. 84.4. 784. If the car's tires had a radius of 1.4 ft, and they...
-
Eminem is a famous hip-hop/rap artist who won a Grammy Award for his music in the movie Eight Mile, in which he starred. His lyrics often contain references to his personal experiences. In "Brain...
-
Grignard reagents undergo a general and very useful reaction with ketones. Methyl magnesium bromide, for example, reacts with Cyclohexanone to yield a product with the formula C 7 H 14 O. What is the...
-
Nitriles, RC N, undergo a hydrolysis reaction when heated with aqueous acid. What is the structure of the compound produced by hydrolysis of propane nitrile, CH3CH2C N, if it has IR absorptions at...
-
Can you explain why analysts might view conference calls/investor presentations as more important than the annual report?
-
Jack and Jill are a married couple living inside a small house. They frequently get drunk and start screaming at each other, causing neighbors to call the police. This is one of those nights, and...
-
A. In details explain the marketing concept with all its components? Apply this concept on an existing product? (Marketing concept are: Consumer Research, Segmentation, Market Targeting,...
-
Carlsville Company began operations in the current year and had no prior stock investments. The following transactions are from it short-term stock investments with insignificant influence. Prepare...
-
Assume a company's activity-based costing system includes three activities with the following activity rates: Activity Cost Pool Activity Rate Testing Setups Molding $2 per testing minute $50 per...
-
___________ goods are those that are in accordance with the obligations under the contract. A) Nonconforming B) Conforming C) Future D) Tangible
-
Zeta Safety, Inc., began the year with $15,000 of common stock and $34,000 of retained earnings. On August 5, investors bought $19,000 of additional stock in the business. On October 22, the business...
-
a. What is the cost of borrowing if Amarjit borrows $28 500 and repays it over a four-year period? b. How many shares of each stock would he get if he used the $28 500 and invested equally in all...
-
The solubility of carbon tetrachloride (CCl 4 ) in water at 25 C is 1.2 g/L. The solubility of chloroform (CHCl 3 ) at the same temperature is 10.1 g/L. Why is chloroform almost ten times more...
-
Predict the products of the reactions of the following compounds with chromic acid and also with PCC. (a) Cyclohexanol (b) 1-methylcyclohexanol (c) Cyclopentylmethanol (d) Cyclohexanone (e)...
-
Two products are observed in the following reaction. (a) Suggest a mechanism to explain how these two products are formed. (b) Your mechanism for part (a) should be different from the usual mechanism...
-
Give the structures of the products you would expect when each alcohol reacts with (1) HCI, ZnCI2; HBr; (3) PBr3: (4) P/I2; (5) SOCI2- (a) butan-1-ol (b) 2-methylbutan-2-ol (c) 2,2-dimethylbutan-1-ol...
-
On January 1, Davis issued $800,000 in 10 year, 7% bonds. The bonds were priced to yield 9%. Interest payments are made on June 30 and December 31. Make all necessary entries for 2022 through Dec....
-
Original Schedule Bracket (0) SS $ $ $ 132,900.00 Rate (1) 132,900.00 12.40% 0.00% New Schedule Bracket Rate (0) (1) S - $ 132,900.00 12.40% $ 132,900.00 $ 250,000.00 0.00% SS $ 250,000.00 $...
-
TRUE/FALSE. Write 'T' if the statement is true and 'F' if the statement is false. 1) Low-volume products often require more special handling than high-volume products.. 2) When overhead is properly...

Study smarter with the SolutionInn App


