A laboratory apparatus to measure the diffusion coefficient of vapor-gas mixtures consists of a vertical, small-diameter column
Question:
A laboratory apparatus to measure the diffusion coefficient of vapor-gas mixtures consists of a vertical, small-diameter column containing the liquid phase that evaporates into the gas flowing over the mouth of the column. The gas flow rate is sufficient to maintain a negligible vapor concentration at the exit plane. The column is 150 mm high, and the pressure and temperature in the chamber are maintained at 0.25 atm and 320 K, respectively. For calibration purposes, you've been asked to calculate the expected evaporation rate (kg/h ? m2) for a test with water and air under the foregoing conditions, using the known value of DAB for the vapor-air mixture.
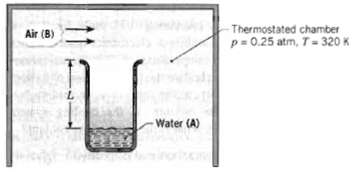
-Thermostated chamber p= 0.25 atm, T-320 K !! Air (B) Water (A)
Step by Step Answer:
KNOWN Column containing liquid phase of water A evaporates into the air B flowing over the mouth of ...View the full answer



Fundamentals of Heat and Mass Transfer
ISBN: 978-0471457282
6th Edition
Authors: Incropera, Dewitt, Bergman, Lavine
Related Video
In this video, the concept of pressure has been explained. The bottle is filled up with water, remove the pipe, and now you can see the clear flow of water from the holes due to liquid pressure.You can see that the second layer is holding the weight of the first layer and the third layer holding the weight of both upper layers, that’s why liquid flows out wider from the third as compared to the first two.At the first hole, water doesn’t roll along the walls; it flows out at an angle.At the second hole, you’ll see that the water flows out at a wider angle as compared to the first hole.At the third hole, we can see that the flow of water is much wider/greater than the other holes.This proves that the pressure at the bottom of the bottle is much more than the pressure at the top layers. P = ????gh
Students also viewed these Mechanical Engineering questions
-
The diffusion coefficient of I in hexane at 25C is 4.05 x 10-9 m2 S-1. Estimate the time required for an iodine molecule to have a root mean square displacement of 1.0 cm.
-
a. The diffusion coefficient of the protein lysozyme (MW = 14.1 kg/mol) is 0.104 10 5 cm 2 s 1 . How long will it take this protein to diffuse an rms distance of 1m? Model the diffusion as a...
-
a. The diffusion coefficient of sucrose in water at 298 K is 0.522 10 9 m 2 s 1 . Determine the time it will take a sucrose molecule on average to diffuse an rms distance of 1 mm. b. If the molecular...
-
Employee earnings records for Redding Company reveal the following gross earnings for four employees through the pay period of December 15. D. Edwards ........... $83,500 A. Seligman...
-
From our frame of reference on Earth, objects slow to a stop as they approach black holes in space because time gets infinitely stretched by the strong gravity near the black hole. If astronauts...
-
1. What are some of the steps that led to creating eHarmony? 2. What advantages did eHarmony have because it was not the first entrant into the online dating market space? 3. What disadvantages might...
-
What should a company do if its rate of return on reinvested earnings is below the weighted average cost of capital?
-
Jason worked various jobs during his teenage years to save money for college. Now it is his twentieth birthday, and he is about to begin his college studies at the University of South Florida (USF)....
-
dy dt 11 Find the indicated derivative. dy fory= (2.5t-5f2)(2t+ 1.2) dt
-
Kashmiri is the largest and most successful specialty goods company based in Bangalore, India. It has not yet entered the North American marketplace, but is considering establishing both...
-
The presence of a small amount of air may cause a significant reduction in the heat rate to a water-cooled steam condenser surface. For a clean surface with pure steam and the prescribed conditions,...
-
Beginning with a differential control volume, derive the diffusion equation, on a molar basis, for species A in a three-dimensional (Cartesian coordinates), stationary medium, considering species...
-
Which of the three agents of transportationwind, water, and icetransports the largest boulders? Why?
-
Olguin Corporation produces a single product and has the following cost structure: Olguin produced 12,000 units during the year and sold 8,000 of these units. There was no beginning inventory. The...
-
Chorio's has computed its Year 2 predictions and has settled on a predetermined overhead application rate of $121.38 per DLH. At the end of this period, actual manufacturing overhead cost for the...
-
Riley Surf Boards is expanding so they are considering building a new factory. John Smith, the company's marketing manager, fully supports building the new factory. Mary Reynolds, the company's chief...
-
Chicago Works employs 2 0 workers. Each employee is paid wages of $ 2 5 per hour and works an 8 - hour workday, Monday through Friday. Employee wages are paid every Friday for the workweek just...
-
Olympic Corporation has 75,000 shares of $1 par value stock outstanding. The largest single stockholder is Lou Cheng, who owns 6,000 shares. On December 31, the total assets of the company amount to...
-
What is the critical test for any accounting system? Does this test rely on syntactic, semantic or pragmatic perspectives?
-
How has the globalization of firms affected the diversity of their employees? Why has increased diversity put an additional burden on accounting systems?
-
Aerosol cans carry clear warnings against incineration because of the high pressures that can develop upon heating. Suppose that a can contains a residual amount of gas at a pressure of 755 mmHg and...
-
Your instructor will provide you with a stock spring suppliers catalog, or pages reproduced from it. Accomplish the task of Prob. 1020 by selecting an available stock spring. (This is design by...
-
The figure shows a conical compression helical coil spring where R1 and R2 are the initial and final coil radii, respectively, d is the diameter of the wire, and Na is the total number of active...
-
A helical coil compression spring is needed for food service machinery. The load varies from a minimum of 4 lbf to a maximum of 18 lbf. The spring rate k is to be 9.5 lbf/in. The outside diameter of...
-
Kenneth Clark is one of Texas's premiere party planners. She purchases a variety of supplies, such as plastic tableware. stemware, and decorations, throughout the year. Kenneth has prepared the...
-
Problem 3-14 (Algo) Schedule of Cost of Goods Manufactured; Overhead Analysis [LO3-3, LO3-4] Gitano Products operates a job-order costing system and applies overhead cost to jobs on the basis of...
-
Required information Use the following information for the Exercises below. [The following information applies to the questions displayed below.] The following adjusted trial balance at December 31...

Study smarter with the SolutionInn App


