Limonene, a fragrant hydrocarbon found in lemons and oranges, is biosynthesized from geranyl diphosphate by the following
Question:
Limonene, a fragrant hydrocarbon found in lemons and oranges, is biosynthesized from geranyl diphosphate by the following pathway. Add curved arrows to show the mechanism of each step. Which step involves an alkene electrophilic addition? (The ion OP2O64- is the diphosphate ion, and ?Base? is an unspecified base in the enzyme that catalyzes the reaction.)
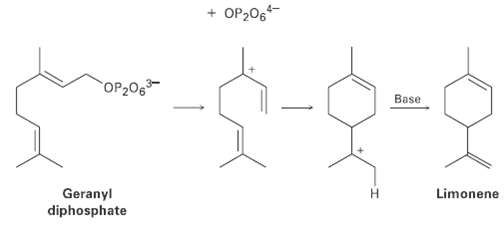
Transcribed Image Text:
+ OP206 OP206 Base Geranyl diphosphate H. Limonene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 92% (14 reviews)
1 2 OP06 3 Base Limonene ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Add curved arrows to show how electrons must move to form the product from the reactants in the following equation, and locate any formal charges. :0 O: HO CH
-
Add curved arrows to the following structures to show how electron pairs must be moved to interconvert the structures, and locate any formal charges. O-H -H
-
Add curved arrows to the following reactions to indicate the flow of electrons for all of the bond-forming and bond-breaking steps. (a) (b) H.
-
The following table describes a randomized trial comparing an experimental medication to a placebo for treatment of reflux. Experimental Treatment Placebo (n = 100) Patient Characteristics (n = 100)...
-
Top-level leaders of major business corporations receive some of the highest compensation packages in the workforce. Why are business leaders paid so much?
-
Suppose the hypothetical processor of Figure 1.3 also has two I/O instructions: 0011 = Load AC from I/O 0111 = Store AC to I/O In these cases, the 12-bit address identifies a particular external...
-
The Luxon Company produces industrial and residential lighting fixtures at its manufacturing facility in Calgary. Shipment of company products to an eastern warehouse is presently handled by common...
-
You purchase a $100,000 life insurance policy for a single payment of $35,000. If you want to earn 9 percent on invested funds, how soon must you die for the policy to have been the superior...
-
Described below are three independent and unrelated situations involving accounting changes. Each change occurs during 2021 before any adjusting entries or closing entries are prepared. a. On...
-
Velky s.r.o. (a Czech company) invests 1,200,000 schillings in a foreign subsidiary on January 1, Year 1. The subsidiary commences operations on that date and generates net income of 400,000...
-
Make sketches of the transition-state structures involved in the reaction of HBr with 1-pentenc (Problem 6.53). Tell whether each structure resembles reactant or product.
-
Epi-Aristolochene, a hydrocarbon found in both pepper and tobacco, is biosynthesized by the following pathway. Add curved arrows to show the mechanism of each step. Which steps involve alkene...
-
Approximately 13% of the general population is left-handed. A researcher has recently come into contact with a number of left-handed artists and wonders whether artists are more likely to be...
-
The following information is provided for the first month of operations for Legal Services Inc.: Prepare a T-accounts to reflect the following business transaction: The Company provided legal...
-
A copper rod is 34.0 m on a winter day when the temperature is -5.0C (Linear expansion coefficient of copper 17 x 10-6/C). How long is the rod on a summer day when the temperature is 38.0C? Express...
-
Pricing Manager, District 6SW From: Vice President, Marketing Re: Strategic Pricing Decision Our (Time Warner's) only competitor is District 6SW currently provides bundled services at $84.95. We are...
-
What's driving the big law firm mergers in Philly and across the U.S.? It was a record year for law firm mergers in 2019, with the announcements by two venerable Philadelphia institutions ranking as...
-
Consider the scenario shown below, with 10 different servers (four shown) connected to 10 different clients. The pairs share a common middle hop with a transmission capacity of R = 200 Mbps. Each...
-
The owner of a garden maintenance shop has been requested to cut and trim a yard. If the owner does the job himself, he estimates that it will take three hours to complete. If he calls a temporary...
-
What kind of financial pressures can an LBO cause?
-
What is the ClausiusClapeyron equation, and why is it important?
-
What is the absolute configuration of (+) -methyl- hexane if catalytic hydrogenation of (S)-(+)-3- methyl-1-hexene gives (-)-3-methylhexane?
-
Draw perspective representation for each of the following chiral molecules, Use models if necessary, (D = deuterium = 2H, a heavy isotope of hydrogen.) (2Z,4R) - 4 - methyl - 2 - hexane
-
Two stereoisomers of the compound (H3N)2Pt(Cl), with different physical properties are known. Show that this fact makes it possible to choose between the tetrahedral and square planar arrangements of...
-
Which type of support is tax-free for federal income tax purposes? Question 62Select one: a. Alimony from a divorce decree entered into March 17th, 2015 b. Child support c. Alimony from a divorce...
-
Place the following steps of the budgeting process in the correct order. Start by choosing the first item in the sequence and clicking, dragging, or using your keyboard to select it. Drag the items...
-
_______ is an employee's final earnings after taxes and other payroll deductions are made. Question 13 options: a) Net pay b) Taxable pay c) Taxable debt d) Assets

Study smarter with the SolutionInn App


