List the point coordinates for all atoms that are associated with the FCC unit cell(figure). (b) (a)
Question:
List the point coordinates for all atoms that are associated with the FCC unit cell(figure).
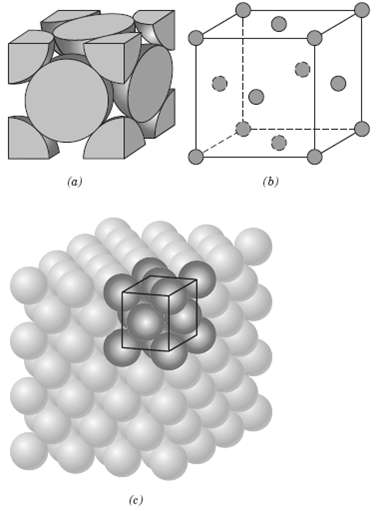
Transcribed Image Text:
(b) (a) (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (12 reviews)
From figure b the atom located of the origin of the unit cell h...View the full answer

Answered By

Dinesh F
I have over 3 years of professional experience as an assignment tutor, and 1 year as a tutor trainee.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
List the point coordinates of all atoms that are associated with the diamond cubic unit cell.
-
List the point coordinates of the titanium, barium, and oxygen ions for a unit cell of the perovskite crystal structure (Figure 12.6).
-
List the point coordinates of the titanium, barium, and oxygen ions for a unit cell of the perovskite crystal structure(figure). Ti4+ Ba2+ 02-
-
An airplane has a mass of 5000 kg, a maximum thrust of 7000 N, and a rectangular wing with aspect ratio 6.0. It takes off at sea level with a 60 split flap as in Fig. 7.25. Assume all lift and drag...
-
Heat a small amount of water to boiling in an aluminum soda-pop can and invert it quickly into a dish of cooler water. Surprisingly dramatic!
-
Canadian partnerships are required to file an annual information return (T5013) containing the following information: a) Income or loss of the partnership, b) Name, address and social insurance...
-
Should family members serve on the same board?
-
Largest Company acquired Large Company on January 1. As part of the acquisition, $10,000 in goodwill was recognized; this goodwill was assigned to Largest's Production reporting unit. During the...
-
K Motorsports of Miami, Inc., reported the following financial statements for 2018: (Click the icon to view the income statement.) comparative balance sheets.) (Click the icon to view the Calculate...
-
Peachtree Delivery Service is owned and operated by Jerome Foley. The following selected transactions were completed by Peachtree Delivery Service during February: 1. Received cash from owner as...
-
Sketch a unit cell for the body-centered orthorhombic crystal structure.
-
List the point coordinates of all atoms that are associated with the diamond cubic unit cell(figure). C
-
Identify and explain the conditions through which goal setting can be used to improve job performance.
-
1010 1. Add the following Chart of Accounts: Bank - Chequing 1600-200 Revenue from Textbook Sales 1030 1 Bank Credit Card 1620-100 Sales Discounts 1100 Accounts Receivable 1620-200 Sales Discounts...
-
Explain the differences among payback period, net present value, and internal rate of return. Which do you think provides the best indicator of financial performance? 2. Explain how the future value...
-
What are visual arts? What types of visual arts are there? What types of artists, art styles and art mediums are there?
-
Describe your experience with this project. What were the challenges you faced with co-planning and co-teaching? How would you rate the contributions of your team members (special educator, general...
-
ZYNE is contemplating acquiring an exploration license for a gold mining location in Western Australia, with plans to extract gold. The license is priced at $10 million, while the expenses for...
-
A fence consists of horizontal and vertical wooden rods with a distance of \(10 \mathrm{~cm}\) between them (measured from the center of the rods). The rods have a circular sectional view with a...
-
What is master production scheduling and how is it done?
-
Let (x) = x 2 + 3x. Calculate R 6 , M 6 , and L 6 for on the interval [2, 5]. Sketch the graph of and the corresponding rectangles for each approximation.
-
The preexponential and activation energy for the diffusion of iron in cobalt are 1.1 10-5 m2/s and 253,300 J/mol, respectively. At what temperature will the diffusion coefficient have a value of 2.1...
-
Self-diffusion involves the motion of atoms that are all of the same type; therefore it is not subject to observation by compositional changes, as with interdiffusion. Suggest one way in which...
-
The diffusion coefficients for iron in nickel are given at two temperatures: (a) Determine the values of D0 and the activation energy Qd. (b) What is the magnitude of D at 1100C (1373 K)? T(K) Ds)...
-
content area Part 1 Yield management is the aggregate planning process of allocating the company's scarce resources to Part 2 A. control low fixed costs. B. maximize revenue. C. stabilize customer...
-
Kyoto Joe Inc. sells earnings forecasts for Japanese securities. Its credit terms are 4/10, net 60. Based on experience, 50% of all customers will take the discount. a. What is the average collection...
-
a.&b. Based upon the size and information provided, select "X" if the programs are Type A with low risk and high risk or Type B with low risk and high risk programs, or select "NA". Program # Award...

Study smarter with the SolutionInn App


