Look at the following electrostatic potential map of chloromethane, and tell the direction of polarization of the
Question:
Look at the following electrostatic potential map of chloromethane, and tell the direction of polarization of the C ? C1 bonds:
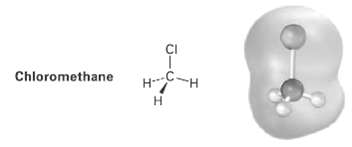
Transcribed Image Text:
CI C-H Chloromethane H.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
In an electrostatic potential map the color red in...View the full answer

Answered By

Ehsan Mahmood
I’ve earned Masters Degree in Business Studies and specialized in Accounts & Finance. Couple with this, I have earned BS Sociology from renowned institute of Pakistan. Moreover, I have humongous teaching experience at Graduate and Post-graduate level to Business and humanities students along with more than 7 years of teaching experience to my foreign students Online. I’m also professional writer and write for numerous academic journals pertaining to educational institutes periodically.
4.90+
248+ Reviews
287+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following electrostatic potential diagrams represent H2, HCl, or NaCl. Label each, and explain your choices. (a) (b)
-
The following electrostatic potential diagrams represent CH4, NH3, or H2O. Label each, and explain your choices. a. C. b.
-
(a) Figure 11.16 is an electrostatic potential map of calicene, so named because its shape resembles a chalice (calix is the Latin word for "cup"). Both the electrostatic potential map and its...
-
A company operates a standard absorption costing system and absorbs fixed production overheads based on machine hours. The budgeted fixed production overheads for the company for the previous year...
-
Paul Barrone is a graduate student at State University. His 10-year-old son, Jamie, lives with him, and Paul is Jamie's sole support. Paul's wife died in 2015, and Paul has not remarried. Paul...
-
To test H0: = 50 versus H1: 50, a simple random sample of size n = 15 is obtained from a population that is normally distributed. The sample mean is 48.1 and the sample standard deviation is 4.1....
-
Fresh Water Testing is considering investing in a new testing device. It has two options: Option A would have an initial lower cost but would require a significant ex- penditure for rebuilding after...
-
Tiger Golf Supplies has $25 million in earnings with 7 million shares outstanding. Its investment banker thinks the stock should trade at a P/E ratio of 31. Assume there is an underwriting spread of...
-
Marine Economics: For the following problem, fill in the blanks. The cost per boat is constant at $2,000. The price per pound is $1,000. a. Given this information complete the following table, pay...
-
Shoe Express, Inc., manufactures two types of shoes, B-Ball and Marathon. The B-Ball shoe has a complex design that uses gel-filled compartments to provide support. The Marathon shoe is simpler to...
-
Use the electro-negatively values shown in Figure to rank the following bonds from least polar to most molar: H3C ? Li, H3C ? K, H3C ? F, H3C ? MgBr, H3C ? OH. 2.1 Li Be 1.0 1.6 Na Mg 0.9 1.2 Ca 0.8...
-
Ethylene glycol, HOCH2CH2OH, has zero dipole moment even though carbon-oxygen bonds are strongly polarized. Explain.
-
A growing number of small and mid-size businesses are enlisting ombudsmen to handle internal conflicts. At Sigmet, a Massachusetts data processor design company, even small coworker disagreements...
-
Read about the case: Dylan Brandt v. Leslie Rutledge, No. 21-2875 (8th Cir. 2022) QUESTIONS TO BE ANSWERED: A. Research Dylan Brandt v. Leslie Rutledge, No. 21-2875 (8th Cir. 2022) and Give a summary...
-
1. Why are cycle time improvements important for long term strategies, and why are value stream maps a useful tool? 2. How does agile project management expose poor work processes or stumbling blocks...
-
GD decides to hedge the market risk with NYMEX gasoline futures each covering 42,000 gallons, using h = 1. 2.1 Is the contract between GD and the gas stations a forwards or a futuresBased on your...
-
Prepare a standard legal memorandum using, for example, the "FILAC" method analyzing the Landlord and Tenant Board matter that I attended and writing about a potential outcome, or if you observed the...
-
The company has a desired operating income of $72,000 per service outlet. Calculate the dollar amount of each type of service that must be provided by each service outlet to meet the company's target...
-
What motivates sales executives and finance and accounting executives to participate in activities that result in inaccurate reporting of revenues?
-
Explain the circumstances that could result in a long-term bank loan being shown in a statement of financial position as a current liability.
-
The metal tin undergoes a transition from a gray phase to a white phase at 286 K and ambient pressure. Given that the enthalpy change of this transition is 2090 J/mol and that the volume change of...
-
Identify the most acidic hydrogen in each of these compounds: a) HOCCH,CH,SOH 0 CO H 0 I e) CHCCHCOCHCH3 0 b) CH-CH,CH,C=N d) 0 CHOH f) HNCHCOH
-
Show the products of these acid-base reactions and predict whether the equilibria favor the reactants or the products: a) CHCCHCCHCH + OCHCH b) CHCHNO + CHO: CH3 (c) CH3COCH, + CHCH- 10 1:Z: LL CH,...
-
Which compound is behaving as the Lewis acid and which as the Lewis base in this reaction? AICI3 T CHCHCHCH3 + AICI CHCHCHCH3
-
Which organization has brought Canada and US to a common format and valuation in reporting the financial statements for the public companies
-
The process of comparing and checking company records against those shown on a bank statement to determine whether the records are in agreement is called what a) bank reconciliation. b) credit audit....
-
6. Cutting bits in a machinery department 7. Food for a factory cafeteria 8. Workers' compensation insurance in a factory 9. Steel scrap for a blast furnace 10. Paper towels for a factory...

Study smarter with the SolutionInn App


