NaHCO3 is decomposed by heat according to If you start with 100.0 g of NaHCO3 and collect
Question:
NaHCO3 is decomposed by heat according to
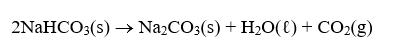 If you start with 100.0 g of NaHCO3 and collect 10.06 L of CO2 over water at 20°C and 0.977 atm, what is the percent yield of the decomposition reaction?
If you start with 100.0 g of NaHCO3 and collect 10.06 L of CO2 over water at 20°C and 0.977 atm, what is the percent yield of the decomposition reaction?
Transcribed Image Text:
2NAHCO3(s) → Na2CO3(s) + H20(t) + CO2(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 45% (11 reviews)
Here we have a stoichiometry problem where we need to find the amount of CO 2 collected when 100...View the full answer

Answered By

John Aketch
I am a dedicated person with high degree of professionalism, particularly in academic writing. My desire is to is to make students excel in their academic endeavor.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physical Chemistry questions
-
NaHCO3 decomposes when exposed to heat: 2NaHCO3(s) ( Na2CO3(s) + CO2(g) + H2O() H = 91.5 kJ What mass of NaHCO3 is decomposed by 256 kJ?
-
What is the equilibrium composition of a reaction mixture if you start with 0.500 mol each of H2 and I2 in a 1.0-L vessel? The reaction is H2(g) + 12(g)--2HI(g) Kc = 49.7 at 458C
-
Part 1: You run the chemical reaction a. Write the equilibrium-constant expression for the reaction. b. Can you come up with some possible concentrations of C, D, and E that you might observe when...
-
How do complementary assets and social complexity influence a firm's organization?
-
How do the auditors evaluate misstatements in a substantive test?
-
At what percentile is \(x=192\) ? Answer the question about normally distributed data with mean 200 and standard deviation 20. Round percentiles to the nearest whole number and round data values to...
-
understand the negative effects of abuse of power and sexual harassment.
-
Because of the huge fixed cost of running pipes to everyones home, natural gas is a natural monopoly. Suppose demand is Q = 100 P and marginal revenue is MR = 100 2Q. Suppose marginal cost is $20,...
-
If f(theta) = acos(btheta)-1, where a > 0 and b>0, then a true statement is \f
-
A Global private bank is aggressively looking to leverage technology to improve customer experience and reduce operational costs. Over the last few years, it has tied up with at least five startups...
-
HNO3 reacts with iron metal according to Fe(s) + 2HNO3(aq) ( Fe(NO3)2(aq) + H2(g) In a reaction vessel, 23.8 g of Fe are reacted but only 446 mL of H2 are collected over water at 25C and a pressure...
-
What is the pressure in pascals if a force of 3.44104 MN is pressed against an area of 1.09 km2?
-
Aside from what was mentioned in the text, give three examples of where periodically valid timing is used.
-
Methanol may be produced by the reaction of carbon dioxide and hydrogen. CO 2 + 3H 2 CH 3 OH + H 2 O The fresh feed to the process contains hydrogen and carbon dioxide in stoichiometric proportion,...
-
Amidst the complexities of advanced knowledge economies and rapidly evolving technological landscapes, what role do cross-disciplinary collaborations and boundary-spanning initiatives play in...
-
Problem 2: (60 points) The basic dimensions and movement range of each of the joints of the MITSUBISHI RM-501 are shown on the following page. A reference coordinate system which is fixed to the...
-
Compare and contrast different sources of legal advice and support for dispute resolution to make appropriate recommendations to legal solutions.
-
Question 6 yet answ Marked out of 3.00 At year-end, Generous Company has a cash generating unit with the following assets and liabilities at carrying amount: Inventory P240,000 Accounts Receivable...
-
A and B are in partnership sharing profits and losses in the ratio 4:3 and for the last four years they have been entitled to annual salaries of 90,000 and 1,50,000 respectively, but not to interest...
-
Dan and Diana file a joint return. Dan earned $31,000 during the year before losing his job. Diana received Social Security benefits of $5,000. a. Determine the taxable portion of the Social Security...
-
The metallic bonding found in the element gold is such that 1.00 ounce of Au can be flattened into a sheet that is 3.00 10 2 ft 2 . If there are 28.35 g/ounce and 30.48 cm/ft, how thin is this...
-
What is the volume change when 1 mol of graphite (d = 2.26 g/cm 3 ) is converted to diamond (d = 3.51 g/cm 3 )?
-
Why do free-floating liquids in an orbiting spacecraft adopt a spherical shape?
-
Europa Company manufactures only one product. Presented below is direct labor information for November. Standard direct labor hours per unit of product 4 . 2 0 Number of finished units produced 5 , 2...
-
The internal rate of return method is used by Royston Construction Co . in analyzing a capital expenditure proposal that involves an investment of $ 6 2 , 4 0 0 and annual net cash flows of $ 1 5 , 0...
-
Calculate Debt Ratio: Requirements: Given the following figures from a balance sheet: Total Liabilities: $500,000 Total Assets: $1,200,000 Compute the debt ratio. Express the debt ratio as a...

Study smarter with the SolutionInn App


