Name the following alkenes, and predict the products of their reaction with (1) meta-chloroperoxybenzoic acid, (ii) KMnO4
Question:
Name the following alkenes, and predict the products of their reaction with (1) meta-chloroperoxybenzoic acid, (ii) KMnO4 in aqueous acid, and (iii) O3, followed by Zn in aceticacid:
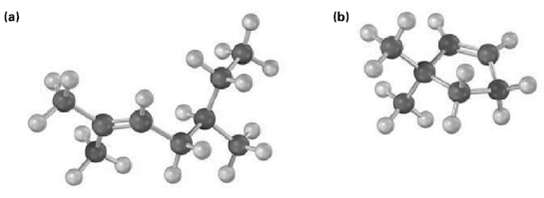
Transcribed Image Text:
(a) (b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
a CH3 CH3CCHCHCHCHCH3 25Dimethyl2heptene CH3 RCO3H KM...View the full answer

Answered By

Vincent Omondi
I am an extremely self-motivated person who firmly believes in his abilities. With high sensitivity to task and operating parameters, deadlines and keen on instructions, I deliver the best quality work for my clients. I handle tasks ranging from assignments to projects.
4.90+
109+ Reviews
314+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Name the following alkynes, and predict the products of their reaction with (i) H2 in the presence of a Lindlar catalyst and (ii) H3O+ in the presence ofHgSO4: (b) (a)
-
Predict the products formed by periodic acid cleavage of the following diols. (a) CH3CH(OH)CH(OH)CH3 (b) (c) (d) CHAOH OH OH Ph-CCH OH)CH,CH CH HO HO
-
Name each of the following alkenes or alkynes. a. CH2 = CH-CH2-CH3 b. c. d. e. f. g. CH3 C-CH-CH3 CH3 CH CH3 CH3CH2CH CH CH CH CH3 CH, C-CH-CH CH, CH2-CH, CH3 CH2CHs CH, CH2CH3 CH3 C C-CH CH3 CH3
-
Consider a process consisting of five resources that are operated eight hours per day. The process works on three different products, A, B, and C; Resource Number of Workers Processing Time for A...
-
Sarpedon Corp. claims that its car batteries average at least 880 CCA (cold-cranking amps). Tests on a sample of 9 batteries yield a mean of 871 CCA with a standard deviation of 15.6 CCA. (a) State...
-
Compare and contrast the American Opportunity Tax Credit with the Lifetime Learning Tax Credit.
-
Create an eulerization using the fewest duplicate edges possible for Graph Z. a b C Graph Z Graph Z d
-
The following are the transactions relating to the formation of Cardinal Mowing Services, Inc., and its first month of operations. Prepare an answer sheet with the columns shown. Record each...
-
8 P 2 - 4 What is the amount of under - applied overhead in Department D ? \ table [ [ A , $ 3 , 0 0 0
-
Personal Electronix sells iPads and iPods. The business is divided into two divisions along product lines. CVP income statements for a recent quarter's activity are presented below. Instructions (a)...
-
One of the chain-termination steps that sometimes occurs to interrupt polymerization is the following reaction between two radicals. Propose a mechanism for the reaction, using fishhook arrows to...
-
Draw the structures of alkenes that would yield the following alcohols on hydration (red = O). Tell in each case whether you would use hydroboration/oxidation oroxymercuration. (b) (a)
-
A company's weekly payroll amounts to $50,000 and payday is every Friday. Employees work five days per week, Monday through Friday. The appropriate journal entry was recorded at the end of the...
-
How do microbial communities shape host physiology and immune function, and what are the implications of dysbiosis in the microbiota for human health, particularly in the context of chronic...
-
a) Discuss the 5 ways to finance the international trade receivables, which is also common ways to finance the trade for a short-term period. b) What are the 3 types of relationship between the...
-
A 249kg piano is pushed & pulled up a ramp into a truck. If the ramp is 5.50m long and the back of the truck is 86cm above ground. How much force are the three men applying to push/pull the piano up...
-
Whitney has a $50,000 outside tax basis in the VWX partnership. Whitney receives the following assets in a current (nonliquidating) distribution: Distributed TB FMV Cash 10,000 10,000 Accts....
-
Variable costing system Product X Product Y Product Z Total k k k k Sales 100k units at 15 1,500 80k units at 25 2,000 120k units at 10 1,200 4,700 Total variable costs 1000 1200 1320 3520...
-
Answer the following questions using the tabulated PERT project data. a. Draw the AON CPM network for the project, perform the forward and backward computations, and determine the project duration....
-
Prove the result that the R 2 associated with a restricted least squares estimator is never larger than that associated with the unrestricted least squares estimator. Conclude that imposing...
-
What happens to the vapor pressure of a substance when its surface area is increased at constant temperature? (a) The vapor pressure increases. (b) The vapor pressure remains the same. (c) The vapor...
-
A student, Flick Flaskflinger, in his twelfth year of graduate work, needed to prepare ethylmagnesium brdmide from ethyl bromide and magnesium, but found that his laboratory was out of diethyl ether....
-
When sec-butylbenzene undergoes free-radical bromi-nation, one major product is formed, If the starting material is optically active, predict whether the substitution product should also be optically...
-
Three alkyl halides, each with the formula C7HBr, have different boiling points. One of the compounds is optically active. Following reaction with Mg in ether, then with water, each compound gives...
-
Oliver was hired as the Chief Technology Officer (CTO) of Delta Corporation, a privately-owned company in the telecommunications sector, effective February 1, 2022. Throughout the fiscal year 2023,...
-
Calculate Return on Equity (ROE): Requirements: Using the following figures: Net Income: $100,000 Average Equity: $500,000 Compute the return on equity (ROE). Express ROE as a percentage. Provide the...
-
Budgeting at Pfizer Values: Item Budgeted Amount Actual Amount Sales $120,000,000 $115,000,000 Variable Costs $60,000,000 $60,000,000 Fixed Costs $40,000,000 $40,000,000 Requirements: Prepare a...

Study smarter with the SolutionInn App


