Name the following amines, and identify each as primary, secondary, ortertiary: (b) (a) (c)
Question:
Name the following amines, and identify each as primary, secondary, ortertiary:
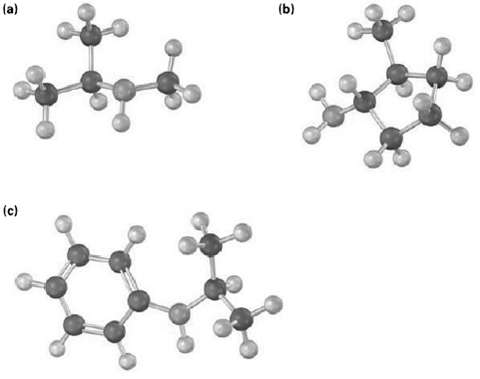
Transcribed Image Text:
(b) (a) (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
a H3C H HC CH3 b H NMethylisop...View the full answer

Answered By

Jonas Araujo
I have recently received the degree of PhD. In Physics by the Universidade Federal do Maranhão after spending a term in Durham University, as I have been awarded a scholarship from a Brazilian mobility program. During my PhD. I have performed research mainly in Theoretical Physics and published works in distinguished Journals (check my ORCID: https://orcid.org/0000-0002-4324-1184).
During my BSc. I have been awarded a scholarship to study for a year in the University of Evansville, where I have worked in detection-analysis of photon correlations in the the Photonics Laboratory. There I was a tutor in Electromagnetism, Classical Mechanics and Calculus for most of that year (2012).
I am very dedicated, honest and a fast learner, but most of all, I value a job well done.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Primary or secondary amines will attack epoxides in a ring-opening process: For substituted epoxides, nucleophilic attack generally takes place at the less sterically hindered side of the epoxide....
-
Primary amines can be converted into secondary amines by reaction with halo alkanes. This reaction is difficult to achieve in the lab because of the large number of byproducts. Select the possible...
-
Which amines in Practice Problem 2.16 are (a) Primary amines, (b) Secondary amines, (c) Tertiary amines? Problem 2.16 (a) (b) (c) (d) Me Z: IN
-
Make a comparison of the following insurance coverage premium bids and determine from a costing standpoint, which offer is better. Show your calculations: Assume that you, as a risk manager would...
-
What qualities of Netflix support the idea that it is a high-performance work system? What other qualities would contribute to it being a high-performance work system?
-
Diameters (cm), circumferences (cm), and volumes (cm3) from balls used in different sports are listed in the table below. Is there sufficient evidence to conclude that there is a linear correlation...
-
Decentralization and Transfer Pricing The city of Medina Park operates a plumbing and electrical maintenance department, responsible for maintaining all water and electric service functions in...
-
On January 1, 2014, the ledger of Shumway Company contains the following liability accounts. Accounts Payable ........... $52,000 Sales Taxes Payable ........... 5,800 Unearned Service Revenue...
-
Carmino Company is considering an investment in equipment that is expected to generate an after-tax income of $7,000 for each year of its four-year life. The asset has no salvage value. The firm is...
-
Today is May 17, 2023, and you, CPA, are a Senior Tax Associate at Borden & Laurier LLP. Sylvie Condurache, Tax Manager, has called you into her office to discuss a long-time client: "I just got out...
-
Compound A, C 6 H 12 O, has an IR absorption at 1715 cm ?1 and gives compound B, C6H15N, when treated with ammonia and NaBH3CN. The IR and 1 H NMR spectra of B are shown. What are the structures of A...
-
The following compound contains three nitrogen atoms. Rank them in order of increasing basicity.
-
Beach Wind Company manufactures kites that sell for $20 each. Each kite requires 2 yards of lightweight canvas, which costs $0.60 per yard. Each kite takes approximately 30 minutes to build, and the...
-
Arrange the following functions into increasing order; that is, f(n) should come before g(n) in your list if and only if f(n) is O(g(n)). (3 pts) 201000, 2010n, n2.01, 0.002n, log log n, nlog n, (lg...
-
We can make a directed equivalent of the random graph by taking n nodes and placing directed edges with probability p between every pair of distinct nodes. We explicitly and separately consider...
-
A security assessor is trying to set up automated scans that check against a predetermined security baseline that checks for vulnerabilities. Which of the following should they set up for this?...
-
First, you need to get your hands on the relevant data. The shipping department has been kind enough to provide you with a repository containing all of their spreadsheets, as well as a copy of the...
-
A city of size 4 2 km 2 is to be covered by a digital cellular phone network. The spectrum re - use cluster size is 3 cells and each cell has an area 0 . 2 km 2 . Assume that the cells perfectly fit...
-
Air in the boundary layer a. has zero velocity at the wing surface. b. is turbulent near the leading edge. c. is more apt to stall if it is turbulent. d. None of the above
-
State whether each statement is true or false. If false, give a reason. {purple, green, yellow} = {green, pink, yellow}
-
At 650 K, the reaction MgCO 3 (s) MgO(s) + CO 2 (g) has K p = 0.026. A 10.0-L container at 650 K has 1.0 g of MgO(s) and CO 2 at P = 0.0260 atm. The container is then compressed to a volume of 0.100...
-
When a solution of salt (sodium chloride) in water is treated with a silver nitrate solution, a white precipitate forms immediately. When tetra chloromethane is shaken with aqueous silver nitrate, no...
-
For each of the following elements, determine (1) how many valence electrons it has and (2) what its common valence is: a. N b. C c. F d. O e. P f. S
-
Write a structural formula for each of the following compounds, using a line to represent each single bond and dots for any unshared electron pairs: a. CH3OH b. CH3CH2Cl c. C3H8 d. CH3CH2NH2 e. C2H5F...
-
Mahogany Company manufactures computer keyboards. The total cost of producing 15,000 keyboards is $430,000. The total fixed cost amounts to $130,000. Determine the total cost of manufacturing 25,000...
-
Assets Liabilities Common Stock Shareholders' Equity Retained Earnings $ In May, Rec Rooms, Inc., collected $8,000 owed by customers for services performed in April. Show the effect of this...
-
is federal legislation providing rules for how private-sector organizations collect, use, or disclose information about employees in the course of for-profit commercial activities in Canada. A)...

Study smarter with the SolutionInn App


