Name the following compound, identify each substituent as axial or equatorial, and tell whether the conformation shown
Question:
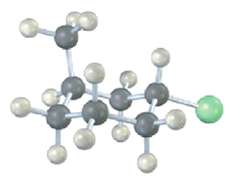
Name the following compound, identify each substituent as axial or equatorial, and tell whether the conformation shown is the more stable or less stable chair form (yellow-greenC1):
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
aCH3 H H CCI ring flip HCe H H tra...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The most stable conformation of 1,3-dioxan-5-ol is the chair form that has its hydroxyl group in an axial orientation. Suggest a reasonable explanation for this fact. Building a molecular model is...
-
Identify each substituent in the following compound as axial or equatorial, and tell whether the conformation shown is the more stable or less stable chair form (yellow green ? CI):
-
The most stable conformation of trqans-l,2-cyclohexanediol is the chair in which both hydroxy groups are equatorial, (a) Draw the structure or, better yet, make a model of the compound in this...
-
Why does allocating an array of length \(n\) take time proportional to \(n\) ?
-
Assume you purchased a high-yield corporate bond at its current market price of $850 on January 2, 2004. It pays 9 percent interest and it will mature on December 31, 2013, at which time the...
-
Refer to Data Set 16 in Appendix B and find the maximum and minimum earthquake magnitudes, then use those values with the range rule of thumb to estimates. How many earthquakes must you randomly...
-
E4-7 Andrea Boss, Inc. manufactures five models of kitchen appliances at its Vista plant. The company is installing activity-based costing and has identified the following activi- ties performed at...
-
The financial statements at the end of Hamel Realtys first month of operations are shown below. Instructions 1. Would you classify a realty business such as Hamel Realty as a manufacturing,...
-
Select the formula, then enter the amounts and compute the standard variable overhead allocation rate. Standard variable overhead allocation rate
-
Give IUPAC name for the followingcompounds: (c) NC. CH 2 () (a) CHCH2CH2CH CH2 CH () CH CH2CO2H (d) (e) CHH2H2H2CH3 CHCN CH (g) Br (h) CN BRCH2CHCH2CH2CO2H
-
Name the following cycloalkanes: (a) (b)
-
A trisubstituted cyclohexane with three substituents? red, yellow, and blue?undergoes a ring-flip to its alternative chair conformation. Identify each substituent as axial or equatorial, and show the...
-
Define the following life insurance terms: beneficiary, face amount, insured, policyholder, and policy owner. How are these terms related?
-
What are the first three points of time (after t = 0), in which the voltage evaluates to 3.0 V, as the function for the voltage is given by u(t) = 4.2 V sin(1.2t - 0.5)? Please give your answers with...
-
A metallurgist has one alloy containing 35 % titanium and another containing 64 % titanium. How many pounds of each alloy must he use to make 52 pounds of a third alloy containing 38 % titanium?...
-
A College Alcohol Study interviewed an SRS of 1000 college students about their drinking habits. Suppose that half of all college students "drink to get drunk" at least once in a while. That is...
-
The ancient Greek mathematician Euclid is credited with the development of the theorem that the sum of the angles of a triangle is 180 degrees. Use the previous information to solve for the measure...
-
You are running the bond trading desk of a large investment bank. This morning, you obtained the following quotes from your broker: There is a 0.5-year maturity zero-coupon bond with price 97.58...
-
Which of the following assurances is not provided by an unqualified opinion on a SysTrust report? a. There are procedures to protect the system against unauthorized physical access. b. The financial...
-
With your classmates, form small teams of skunkworks. Your task is to identify an innovation that you think would benefit your school, college, or university, and to outline an action plan for...
-
Butanol and pentane have approximately the same mass, however, the viscosity (at 20 C) of butanol is = 2.948 cP, and the viscosity of pentane is cP. Explain this difference.
-
When the 3-bromo-2-butanol with the stereochemical structure A is treated with concentrated HBr, it yields meso-2,3-dibromobutane; a similar reaction of the 3-bromo-2-butanol B yields...
-
Reaction of an alcohol with thionyl chloride in the presence of a tertiary amine (e.g., pyridine) affords replacement of the OH group by Cl with inversion of configuration (Section 11.9). However, if...
-
Draw all of the stereoisomers that are possible for 1,2,3-cyclopentanetriol. Label their chirality centers and say which are enantiomers and which are diastereomers. (Some of the isomers contain a...
-
Blossom Fiber Company is the creator of Y-Go, a technology that weaves silver into its fabrics to kill bacteria and odor on clothing while managing heat. Y-Go has become very popular in undergarments...
-
Supongamos que el estado de flujos de caja de una empresa muestra un aumento de la tesorera. Cul de las siguientes transacciones podra contribuir al aumento de efectivo? Una disminucin de las cuentas...
-
Required information [The following information applies to the questions displayed below.] In each of the cases below, assume Division X has a product that can be sold either to outside customers or...

Study smarter with the SolutionInn App


