Order each of the following sets of compounds with respect to SN2reactivity: CI CH (a) CH3CH2CHCH3 H
Question:
Order each of the following sets of compounds with respect to SN2reactivity:
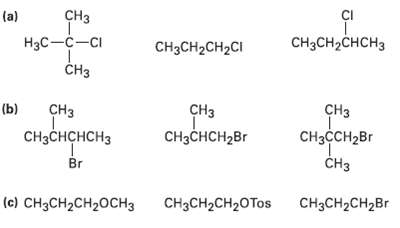
Transcribed Image Text:
CI CHз (a) CH3CH2CHCH3 Hас —с—сі CH3CH2CH2CI CHз (b) CHз CHз CHз CHзCнCH2Br CнзснсHсHз CнзссH2Br Br CHз (c) CH3CH2CH20CH3 CHяCH2CH2Br CHзCH2CH20Tos
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
SN2 reactivity Least reactive a b c CH3 HCCCl CH3 ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Order each of the following sets of compounds with respect to SN1reactivity: H3C CH3 C NH2 (a) i CH3CH2CHCH3 CI CH (b) (CH)3CI (CH)Br (CH)3C (c) Br CHCH3 C-Br CBr 3
-
Order each of the following sets from the least exothermic electron affinity to the most. a. F, Cl, Br, I b. N, O, F
-
Order the compounds in each of the following sets with respect to increasing acidity: (a) Acetic acid, oxalic acid, formic acid (b) p-Bromobenzoic acid, p-nitro benzoic acid, 2, 4-dinitrobenzoic acid...
-
Roberts Originals Co. (ROC) provides new and unique cases and otherassignments to professors each semester to ensure that students will not be able to find the solutions published online. Due to the...
-
Suppose you are an HR managers at a company with high turnover among middle managers (that is, many of them quit to work elsewhere.) Write a brief argument telling the company's executive why a...
-
Define each of the following theories in a sentence or Simple Equation: A. Interest Rate Parity theory B. Expectations theory of forward rates C. Law of one price D. International Fisher Effect...
-
Expected value of research projects Patrick Corporation is preparing its research and development budget for the coming year. Two research projects are being evaluated. Project A is expected to cost...
-
Blanchard Company manufactures a single product that sells for $ 180 per unit and whose total variable costs are $ 135 per unit. The companys annual fixed costs are $ 562,500. (1) Use this...
-
Solano Company has sales of $ 7 4 0 , 0 0 0 , cost of goods sold of $ 4 9 0 , 0 0 0 , other operating expenses of $ 4 6 , 0 0 0 , average invested assets of $ 2 , 2 0 0 , 0 0 0 , and a hurdle rate of...
-
Busy Sally Socialite has trouble remembering people's birthdays, so she has organised her friends into what she calls a Birthday Support Team, or BST. Each friend needs only to keep track of three...
-
The reactions shown below are unlikely to occur as written. Tell what is wrong with each, and predict the actualproduct. OCCH3)3 . H2CH Br (a) CHCH-CH (CH]l3c (b) Na* - CI LCH3 LCH3 (c) Socil,...
-
Predict the product and give the stereochemistry resulting from reaction of each of the following nucleophiles with (R)-2-bromooctane: (a) CN (b) CH3CO2 (c) CH3S
-
Margaret Magee has served both as an outside director to Maxcor Manufacturing since 2007 and as a member of the companys compensation committee since 2011. Margaret has been reviewing Maxcors 2014...
-
Develop a pro-forma financial statement for the next year, noting that coffee inventory will increase by 20% and same store sales will remain flat. ** I am confused on how to do this. Inventory is an...
-
Write on autocratic crime, you need to address (1) what autocratic crime is, its main characteristics and symptoms; (2) what social factors and forces lie under autocratic crime; (3) how autocratic...
-
You borrow money on a self liquidating installment loan (equal payments at the end of each year, each payment is part principal part interest) Loan amount Interest Rate Life Date of Loan $618,000...
-
Compare between Autocratic and Supportive management behavioural models.?
-
Choose a Fortune 5 0 0 Company and review the most recent annual report. Identify the company. Based on the information in the annual report, build a balanced scorecard for your company. Explain why...
-
Spacemonitor plc is building a factory on a site that it already owns. The total investment in the factory (excluding the site) will be 10 million. Management expects the factory to generate new net...
-
Highland Theatre is owned by Finnean Ferguson. At June 30, 2014, the ledger showed the following: Cash, $6,000; Land, $100,000; Buildings, $80,000; Equipment, $25,000; Accounts Payable, $5,000;...
-
Consider the reaction: A solution is made containing an initial [Fe 3 + ] of 1.0 * 10 -3 M and an initial [SCN ] of 8.0 * 10 -4 M. At equilibrium, [FeSCN 2 + ] = 1.7 * 10 -4 M. Calculate the value...
-
Predict the products of saponification of the following esters. (a) (b) (c) (d) CH CHC-OCH-CH
-
Show how you would accomplish the following syntheses in good yields. (a) (b) (c) (d) (e) (f) (g) (h) NH-C-H NH2 COOH C-O-C CH HO COOH COCH(CH3)2 COOH COOH CHO CH2OH CHO COOH COOCH3 CH2OCCH3 COOCH...
-
Propose mechanisms for the following reactions. (a) (b) (c) (d) (e) (f) (g) Does this reaction proceed with retention, inversion, or racemization of the asymmetric carbon atom? Ph-CCI(CH3)2CHOHPhC...
-
Cost-Volume-Profit (CVP) Analysis at Nike Values: Selling Price per Unit: $100 Variable Cost per Unit: $60 Fixed Costs: $1,000,000 Requirements: Calculate the breakeven point in units and dollars...
-
A manufacturing company produces 50,000 units of a product annually. Fixed costs, including rent, utilities, and salaries, total $250,000. Variable costs per unit, including direct materials and...
-
XYZ Corporation's balance sheet on December 31, 2023, is provided below: Item Amount ($) Accounts Payable 1,240,000 Notes Payable 1,220,000 Accrued Expenses 1,100,000 Total Current Liabilities ?...

Study smarter with the SolutionInn App


