Phenol, C6H5OH, is a stronger acid then methanol, CH3OH, even though both contains an O ? H
Question:
Phenol, C6H5OH, is a stronger acid then methanol, CH3OH, even though both contains an O ? H bond. Draw the structures of the anions resulting from loss of H+ from phenol and methanol, and use resonance structures to explain the difference in acidity.
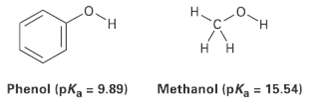
Transcribed Image Text:
H. H. Methanol (pKa = 15.54) = 9.89) Phenol (pK
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
H 66666 HH When p...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Organic Chemistry questions
-
Explain the following observations: (a) HNO3 is a stronger acid than HNO2; (b) H2S is a stronger acid than H2O; (c) H2SO4 is a stronger acid than HSO4-; (d) H2SO4 is a stronger acid than H2SeO4; (e)...
-
Explain the following observations: (a) HCl is a stronger acid than H2S; (b) H3PO4 is a stronger acid than H3AsO4; (c) HBrO3 is a stronger acid than HBrO2; (d) H2C2O4 is a stronger acid than HC2O4-;...
-
Use resonance formulas to explain why polyacetylene has delocalized molecular orbitals extending over the length of the molecule, whereas the following molecule does not. HHHHH
-
Why should one-time write-offs of fixed capital be used in absorption cost systems?
-
Samuel and Annamaria are married, file a joint return, and have three qualifying children. In 2016, they earn wages of $34,000 and no other income. Determine the amount of their earned income credit...
-
Chris LeBlanc estimates that if he does 5 hours of research using data that will cost $75, there is a good chance that he can improve his expected return on a $10,000, 1-year investment from 8% to...
-
Ansara Company sells two products, X and Y. Their unit contribution margins are \($52\) and \($70\). respectively, and their sales mix is 2:1. What is the weighted average unit contribution margin?
-
The following equity investment-related transactions were completed by Lance Company in 2010: Jan. 12. Purchased 1,800 shares of Baxter Company for a price of $56.50 per share plus a brokerage...
-
Sudoku Company issues 33,000 shares of $8 par value common stock in exchange for land and a building. The land is valued at $233,000 and the building at $368,000. Prepare the journal entry to record...
-
1. The university rules allow an F grade to be overridden by any pass grade (A, B, C, D). Now, create a view that lists information about all fail grades that have not been overridden (the view...
-
We?ll see that organic molecules can be classified according to the functional groups they contain, where a functional group is a collection of atoms with a characteristics chemical reactivity. Use...
-
Monobromination of toluene gives a mixture of three bromotoluene products. Draw and name them.
-
What is the attractive feature of a turret-type punch press?
-
Use the following table, Present Value of an Annuity of 1 Period 8% 9% 10% 1 0.926 0.917 0.909 2 1.783 1.759 1.736 3 2.577 2.531 2.487 A company has a minimum required rate of return of 9%. It is...
-
ASD Limited manufactures ovens with a selling price of $1,400. Currently, variable costs are $600 per unit and fixed costs are $100,000 per year. In the year just ended, the company only managed to...
-
What effects will COVID likely have on commercial real estate? If you were a commercial mortgage lender, how might your criteria change for approving office or industrial construction? What industry...
-
How do organizational change efforts reconcile the tension between innovation imperatives and the need for operational stability and continuity ?
-
Custom Chairs Corporation borrowed $10,000 from Town Bank for three years at 10% interest. What is the proper journal entry to record the initial transaction?
-
The following information relates to two companies in the same industry. Required: a. Calculate the percentage growth rate of each companys net earnings from 2004 to 2007. b. Based on this...
-
Write a paper by answer the following question: Should Recycling Be Mandatory?
-
Using the Redlich-Kwong equation of state, compute the following quantities for nitrogen at 298.15 K. a. The difference C P C V as a function of pressure from low pressures to very high pressures b....
-
Arrange these compounds in order of increasing SN2 reaction rate: CI Br CI Br
-
Arrange these compounds in order of increasing SN1 reaction rate: Ph-Br Br + Br CI
-
Show the products of these reactions and explain whether each would follow an SN1 or an SN2 mechanism: a) C) B CI + OH Br + SH DMF CHOH HO Br + HO CHOH HO b) d) f) CI + HO CH,OH OTS + CH0 Br + CHCO...
-
FIFO Perpetual Inventory The beginning inventory at Dunne Co. and data on purchases and sales for a three-month period ending June 30 are as follows: Number Date Transaction of Units Per Unit Total...
-
Question Bl Martina Company is engaged in purchase and sales of leather products. The company adjusts its accounts monthly, closes its accounts annually on 31 December and adopts a perpetual...
-
Which of the following is necessary for a budget to be effective? Goals reflected in a budget should be challenging but attainable. Employees affected by a budget should help prepare it. Managers...

Study smarter with the SolutionInn App


