Predict the product of the following Diels?Alder reaction: ?
Question:
Predict the product of the following Diels?Alder reaction:
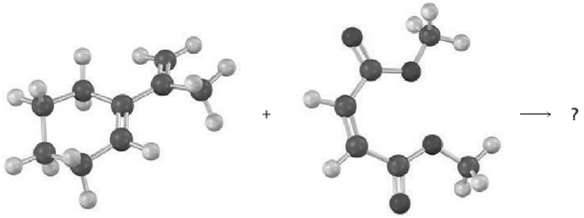
Transcribed Image Text:
?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (17 reviews)
Rotation of the diene to th...View the full answer

Answered By

Ishrat Khan
Previously, I have worked as an accounting scholar at acemyhomework, and have been tutoring busines students in various subjects, mostly accounting. More specifically I'm very knowledgeable in accounting subjects for college and university level. I have done master in commerce specialising in accounting and finance as well as other business subjects.
5.00+
138+ Reviews
432+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the product of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) HBr (excess), OH (1) NaH OH Br2 (excess) H20 H3C OH excess QH (1) NaH,Br (2) heat HNO3, H2SO4 NaNH2, NHa (1) NaOH...
-
Predict the product of the following reaction. O (1) BrMg (2) H2O MgBr (1 equiv.)
-
Predict the product of the reaction if CH3-S-CH2CH2CH2CH2-Br is heated in a polar organic solvent, such as methanol. Similarly, what would be the product for hexyl bromide in methanol? (In hexyl...
-
Dixie Irwin is the department manager for Religious Books, a manufacturer of religious books that are sold through Internet companies. Irwins bonus is based on reducing production costs. Irwin has...
-
Why is infrastructure essential to economic development?
-
Brookman uses three departments to produce plastic handles for screwdrivers: Mixing, Moulding, and Drying. The Assembly Department attaches the screwdriver shanks to the handles. Brookmans Drying...
-
Compute the future value of \($1,000\) continuously compounded for a. 5 years at a stated annual interest rate of 12 percent. b. 3 years at a stated annual interest rate of 10 percent. c. 10 years at...
-
Deleon Inc. is preparing its annual budgets for the year ending December 31, 2014. Accounting assistants furnish the data shown below. An accounting assistant has prepared the detailed manufacturing...
-
Chris and Jamie divorced in November of Year 1. Chris moved out and Jamie remained in their home with their one-year old son, Boyd. Janice, Jamie's mother, lived in the home and acted as Body's nanny...
-
1. The transition from officer to supervisor is difficult and sometimes isolating. Discuss some issues that complicate the transition. 2. What should you do as a new sergeant to prove to your...
-
Which of the following dienes have an s-cis conformation, and which have an s-trans conformation? Of the s-trans dienes, which can readily rotate tos-cis? (a) (c) (b)
-
Draw a segment of the polymer that might be prepared from 2-phenyl-1, 3-butadiene.
-
The following information is available for Stamos Corporation for the year ended December 31, 2017. Beginning cash balance .............................................. $ 45,000 Accounts payable...
-
If f(x) = 2x-4 what is its inverse function, f (x)?
-
As Amazon's sales and corresponding shipping volume increase, there is increased pressure on logistics. If there is no proportional increase in staffing and machinery nor an exogenous advance in...
-
Oscar's Red Carpet Store maintains a checking account with Academy Bank. Oscar's sells carpet each day but makes bank deposits only once per week. The following provides information from the...
-
Online Reviews are often the first step of a customer's experience. Because of that, it is important to track online reviews, and take the good with the bad. You have been hired as a Manager at a...
-
In the following diagram, let V= 0 at infinity. 12.0 cm 12.0 cm 91 + H +8.00- cm 4.00 cm -7.90 nC. a b HJ 92 4.00 cm where 91 = +7.90 nC and 92 = What is the change in electric potential energy if a...
-
The non-symmetrically laminated beam shown in Figure 7.71 consists of a substrate material having Young's modulus \(E_{\mathrm{S}}\) and a coating material having Young's modulus \(E_{\mathrm{C}}\),...
-
Consider the following cash flows in Table P5.5. (a) Calculate the payback period for each project. (b) Determine whether it is meaningful to calculate a payback period for project D. (c) Assuming...
-
What is the effect on vapor pressure of a solution with particularly strong solutesolvent interactions? With particularly weak solutesolvent interactions?
-
(a) Use the polygon rule to draw an energy diagram (as in Figures 16-5 and 16-7) for the MOs of a planar cyclooctatetraenyl system. In Figure 16.5 In Figure 16.7 (b) Fill in the eight pi electrons...
-
Step 2 of the iodination of benzene shows water acting as a base and removing a proton from the sigma complex. We did not consider the possibility of water acting as a nucleophile and attacking the...
-
In an aqueous solution containing sodium bicarbonate, aniline reacts quickly with bromine to give 2, 4, 6-tribromoaniline. Nitration of aniline requires very strong conditions, however, and the...
-
Your video analysis of the motion of a marble gives it position in frame 23 as = (x23, 923) (0.134 m, 0.120 m) and its position in frame 24 as (x24, 24) = (0.102 m, 0.112 m). You estimate that you...
-
Compute the unit binormal vector and torsion of the curve. 9) r(t)=(6 sint, 6 cost, -t) 9) A) B(t) = (cost,-sint, -6) 37 T = B) B(t)= (-cost, sint, -1) T = C) B(t): (-sint, cost, -6) 37 1 T = D)...
-
which driver weakened the swiss franc? a) a surprise change in inflation expectations b) a surprise change in valuation expectations c) a surprise change in interest rates expectations d) a surprise...

Study smarter with the SolutionInn App


