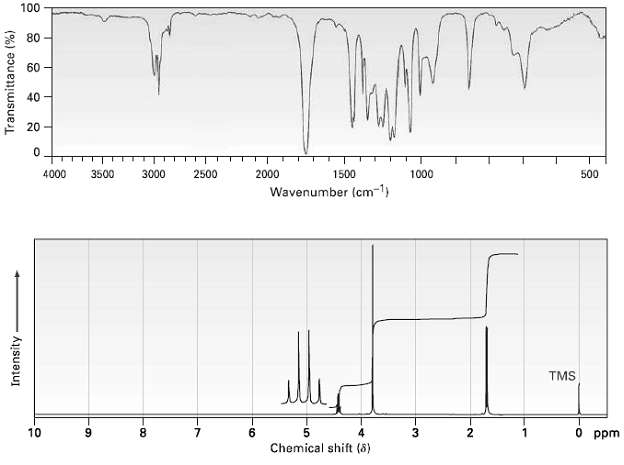
Propose a structure for a compound, C4H7ClO2 that has the following 1R and 1H NMRspectra: 100 80
Question:
Propose a structure for a compound, C4H7ClO2 that has the following 1R and 1H NMRspectra:
Transcribed Image Text:
100 80 60 40 20 4000 3000 2500 2000 1000 500 3500 1500 Wavenumber (cm-1) TMS O ppm 10 6. Chemical shift (8) Transmittance (%) Intensity -08- -LO
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
The IR spectrum indic...View the full answer

Answered By

Chiranjib Thakur
I have no tutoring experience yet, but I can share my skills and knowledge gained from my education and work experiences. I have been a CPA since 2012 with 6 years of work experience in internal auditing and 4 years of work experience in accounting at the supervisory level.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a structure for a compound with molecular formula C 10 H 14 O that exhibits the following 1 H NMR spectrum. Proton NMR Chemical shift (ppm)
-
Propose a structure for a compound with molecular formula C 3 H 8 O that exhibits the following 1 H NMR and 13C NMR spectra: Proton NMR 0.5 5.0 4.5 4.0 3.5 3.0 2.0 1.5 1.0 25 Chemical shift (ppm)...
-
Propose a structure for a compound with molecular formula C 8 H 10 O that exhibits the following 1 H NMR spectrum: Proton NMR Chemical shift (ppm)
-
IKEA was founded in 1943 by a 17-year-old Swede named Ingvar Kamprad. The company, which initially sold pens, Christmas cards, and seeds from a shed on Kamprad?s family farm, eventually grew into a...
-
How do report writers decide what type of graphic to use in a report?
-
Three Statistics classes all took the same test. Here are histograms of the scores for each class. a) Which class had the highest mean score? b) Which class had the highest median score? c) For which...
-
What assumptions must be made for inverse P/E to provide an approximate estimate of required rate of return?
-
Given Targets current situation, what recommendations would you make to Steinhafel for his companys future? When you hear the term discount retail, two names that usually come to mind: Walmart and...
-
of 4 Book Hint Print ferences Required information Use the following information for the Quick Study below (Static) (15-18) [The following information applies to the questions displayed below.] Trey...
-
Five weeks ago, Robin Corporation borrowed from the commercial finance company that employs you as a loan officer. At that time, the decision was made (at your personal urging) to base the loan rate...
-
How would you distinguish spectroscopically between the following isomer pairs? Tell what differences you would expect to see. (a) N-Methylpropanamide and N, N-dimethylacet amide (b)...
-
Assign structures to compounds with the following 1H NMR spectra:? (a) C 4 H 7 ClO? ??IR: 1810 cm ?1 ? (b) C 5 H 7 NO 2 ? ?IR: 2250, 1735 cm ?1 ? (c) C 5 H 10 O 2 ? ? ?IR: 1735 cm ?1 ? TMS O ppm 10...
-
What is the relationship between the revenue and expense accounts and the owners equity account?
-
Suppose that, as a result of a public health crisis, commercial banks decide to increase the amount of cash in their vaults by $200m, in case this is needed to meet a sudden outflow of deposits. (a)...
-
How often is the annual inventory turnover for items like potatoes, corn, oil and seed, which are all perishable?
-
How does finite element analysis contribute to the structural integrity assessment of complex civil engineering projects ?
-
1 How often must HEPA filters be certified? 2 Where would oxycodone be stored in the pharmacy?
-
According to the movie "The Big Short", answer the following questions: 1) what the commercial banks were supposed to do; 2) what commercial banks actually did; 3) what the consequences are; 4)...
-
A \(\$ 100,000\) asset is depreciated for six years on a straight-line basis using a 10 -year life and a \(\$ 10,000\) residual value. In year 7 , the remaining life is changed to five years, with a...
-
Rewrite Programming Exercise 7.5 using streams. Display the numbers in increasing order. Data from Programming Exercise 7.5 Write a program that reads in 10 numbers and displays the number of...
-
What is an antibonding molecular orbital?
-
Using the information in Table 16.2, predict the product(s) of Table 16.2 Friedel-Crafts alkylation of a large excess of ethylbenzene with chloromethane in the presence of A1C13. Eq. 16.23 TABLE 16.2...
-
Using the information in Table 16.2, predict the product(s) of Table 16.2 Friedel-Crafts alkylation of a large excess of ethylbenzene with chloromethane in the presence of A1C13. Eq. 16.23 TABLE 16.2...
-
Draw the carbocation that results from the reaction of the electrophile at the ortho position of anisole; show that this ion also has four resonance structures.
-
a. Net income was $471,000. b. Issued common stock for $78,000 cash. c. Paid cash dividend of $18,000. d. Paid $115,000 cash to settle a long-term notes payable at its $115,000 maturity value. e....
-
Cul es la parte imponible de la pensin de Tara de Oak Enterprises utilizando el mtodo simplificado? a. 0 dlares b. 17.862 dlares c. 18.035 dlares d. 18.485 dlares
-
10-4 (Algo) Computing Issue Prices of Bonds Sold at Par, at a Discount, and at a Premium LO10-2, 10-4, 10-5 James Corporation is planning to issue bonds with a face value of $501,000 and a coupon...

Study smarter with the SolutionInn App


