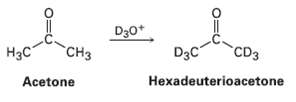
Reaction of acetone with D 3 O + yields hexadeuterioacetone. That is, all the hydrogen?s in acetone
Question:
Reaction of acetone with D3O+ yields hexadeuterioacetone. That is, all the hydrogen?s in acetone are exchanged for deuterium. Review the mechanism of mercuric ion?catalyzed alkyne hydration, arid then propose a mechanism for this deuterium incorporation.
Transcribed Image Text:
D30* "CHз H3C D3C CD3 Acetone Hexadeuterioacetone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (17 reviews)
24txA4 HC HC HC CH3 enol CH3 CH3 CH3 HC HCC HC ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The mechanism involved in the reaction catalyzed by phosphoglyceromutase is known to involve a phosphory-lated enzyme intermediate. If 3-phosphoglycerate is radioactively labeled with 32P, the...
-
Deuterium (D, or 2H; is an isotope of hydrogen with atomic mass - 2. Deuterium can be introduced into organic compounds by using reagents in which hydrogen has been replaced by deuterium. Outline...
-
The hydration of an alkyne is not a reasonable preparative method for each of the following compounds. Explain why. (CH)C C CH
-
Kevin Steven opened a small tax-preparation service. Steven Tax Services trial balance at the end of its second year of operation is as follows. The following information is also available: a. Office...
-
Hill-O-Beans Coffee Company blends four component beans into three final blends of coffee: one is sold to luxury hotels, another to restaurants, and the third to supermarkets for store label brands....
-
Owl, Inc., was owned entirely by Jeri Bell and Jerry Gore, each owning 620,000 of the 1,240,000 shares of common stock outstanding. On January 1, 2015, Owl established an employee stock ownership...
-
Your business uses the allowance method to account for uncollectible receivables. At the beginning of the year, Allowance for Uncollectible Accounts had a credit balance of \($1,100.\) During the...
-
The supply curve for product X is given by Qsx = - 520 + 20Px. a. Find the inverse supply curve. b. How much surplus do producers receive when Qx = 400? When Qx = 1,200?
-
The Major players in the case were Estelle T. Griswold, who was the executive director of Planned Parenthood in Connecticut at the time, and the State of Connecticut. The root of the conflict goes...
-
You intend to install a gas system in your company's car. Use known investment evaluation methods (using the time value of money) and financial assumptions (additional costs, savings, changes in...
-
A cumulene is a compound with three adjacent double bonds. Draw an orbital picture of a cumulene. What kind of hybridization do the two central carbon atoms have? What is the geometric relationship...
-
Give IUPAC names for the following alkylhalides: (b) (a) CHCH2CH2CH21 H C (c) C,2I BrCH2CH2CH2CH2Br CH (e) Br (1) 1 -CH2CI CI CH (d) CH2H2CI CHH2H CHCH2CH2CH I
-
Using the results of the classic Haire and associates study as a basis for your answer, compare and contrast managers' attitudes toward leadership practices in Nordic-European and Latin-European...
-
To view the human economy in isolation from the natural environment is not only misguided but also dangerous. Comment.
-
Identify the following: (a) The three distinctive contributions of the natural environment to the human economy. (b) The three roles of social institutions in a market economy.
-
Consider the model in section 5.3. (a) Derive the firms first-order conditions for profits maximization when they are subject to the relative standard given by Eq. (5.45). (b) Derive a rule for the...
-
Derive a formula for the second-best relative standard to regulate a polluting monopolist.
-
Show that the monopoly output and emissions determined by (6.12) are decreasing in the tax rate. That is, verify equations (6.17) and (6.18). Equation 6.12 Equation 6.17 Equation 6.18...
-
An attorney sees a plastic surgeon and is so happy with her face-lift that she begins to refer all her friends and family. At her 6-month follow-up, she says, So, Doc, Ive sent you all these...
-
Which, if any, of the dichloroethene molecules drawn in Data Table II (3.) (4.) and (5.) are geometric isomers? A. B. C. D. cis-1,2-dichloroethene and trans-1,2-dichloroethene...
-
What is a gas-evolution reaction? Give an example.
-
Give the relationships between the following pairs of structures. The possible relationships are: same compound, cis-trans isomers, constitutional (structural) isomers, not isomers (different...
-
Sulfur dioxide has a dipole moment of 1.60 D. Carbon dioxide has a dipole moment of zero, even though C-O bonds are more polar than S-O bonds. Explain this apparent contradiction.
-
For each of the following compounds, 1. Draw the Lewis structure. 2. Show how the bond dipole moments (and those of any nonbonding pairs of electrons) contribute to the molecular dipole moment. 3....
-
Examine the existential themes in Albert Camus's "The Stranger." How does the protagonist's sense of detachment and absurdity illustrate existential philosophy, and what does this imply about the...
-
Peter Finch is the managing director of Dolls House Boutique Pty Ltd, a small, family-owned company that manufactures timber dolls houses. The company has a couple of standard designs that are very...
-
All business in the HT&E industry are required to comply with the legislation listed below. Your task is to elaborate further into how these Laws apply to your industry and 'HOW' your workplace...

Study smarter with the SolutionInn App


