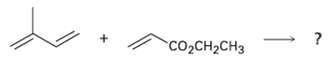
Reaction of isoprene (2-methyl-1, 3-hutadiene) with ethyl propenoate gives a mixture of two Diels?Alder adducts. Show the
Question:
Reaction of isoprene (2-methyl-1, 3-hutadiene) with ethyl propenoate gives a mixture of two Diels?Alder adducts. Show the structure of each, and explain why a mixture is formed.
Transcribed Image Text:
CO2CH2CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
X Joyl OCHCH3 X H H H OCHCH3 i ...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following cycloalkene gives a mixture of two alcohols on hydroboration followed by oxidation. Draw the structures of both, and explain the result.
-
Define expected cash flows, and explain why this concept is important in evaluating projects.
-
Define socialization and explain why this process is so important; explain Sigmund Freud's views as they relate to our individual desires and the demands of society; identify the ways in which we...
-
Split the number 750 into two numbers x and y so that the sum of 8% of the first number and 24% of the second number is 11.2% of the sum of x and y.
-
Google is a company that embraces innovation. What are some actions Google takes to ensure they are creating an environment that supports innovation?
-
Summarized balance sheets of Corner Company and its subsidiary Brook Corporation on December 31, Year 4, were as follows: On the date that Corner acquired its interest in Brook, there was no...
-
What is the value of a call option if the underlying stock price is $20, the strike price is $22, the underlying stock volatility is 50 percent, and the risk-free rate is 4 percent? Assume the option...
-
Flex Displays designs and manufactures displays used in mobile devices. Serious flooding throughout North Carolina affected Flex Displays facilities. Inventory was completely ruined, and the companys...
-
Given below is a portion of Swinging Tunes, Inc.'s management performance report: Contribution margin Controllable fixed costs Budget Actual Difference $1,040,000 $1,020,000 $20,000 430,000 420,000...
-
How high h must the light bulb in Figure 5 be if the point (1, 25, 0) is on the edge of the illuminated region? Light hu b
-
Would you expect a conjugated diyne such as 1, 3-butadlyne to undergo DielsAlder reaction with a dienophile explain.
-
Rank the following dienophiles in order of their expected reactivity in the Diels?Alder reaction. CH CH NC NC CN c=C CH c=c c=C c=C NC CN
-
Distinguish between 1. Job costing; 2. Process costing; and 3. Batch costing. Fully explain each approach.
-
The total budget for launching a credit card is $1,000,000. New product targets customer from z generation and it will be distributed visa call centre and company website. one deliverable is call...
-
Discuss the concept of genetic linkage and how it relates to the physical distance between genes on chromosomes, as well as its significance in genetic mapping and understanding patterns of...
-
You are a sales representative of an Estonian company and your job is to broker domestic wild berries to the UK market. You have just reached an agreement to deliver 60 000 kg of berries to UK at a...
-
The Nantucket Nugget's current debt-to-equity ratio is 40% and pays no taxes. The firm currently has $4 million worth of debt outstanding. Nantucket is considering increasing its debt-to-equity ratio...
-
A company invested in a material handling equipment for $250,000. The installation costs incurred were $50,000. The material handling equipment has a MACRS-GDS 5-year property class. The BT&LCF...
-
The IT manager of Aztec Realty, Inc., prepared the balance sheet of the company while the accountant was ill. The balance sheet contains numerous errors. In par ticular, the IT manager knew that the...
-
Suppose that A is an m n matrix with linearly independent columns and the linear system LS(A, b) is consistent. Show that this system has a unique solution.
-
A solution contains equal amounts (in moles) of liquid components A and B. The vapor pressure of pure A is 100 mmHg and that of pure B is 200 mmHg. The experimentally measured vapor pressure of the...
-
(a) Outline the syntheses indicated in Solved Problem 18-2, beginning with aldehydes and alkyl halides. (b) Both of these syntheses of 1-phenylbuta-1, 3-diene form the central double bond. Show how...
-
Show how Wittig reactions might be used to synthesize the following compounds. In each case, start with an alkyl halide and a ketone or an aldehyde. (a) Ph-CH==C(CH3)2 (b) Ph-C(CH3)==CH2 (c)...
-
Propose mechanisms for (a) The acid-catalyzed hydration of chloral to form chloral hydrate. (b) The base-catalyzed hydration of acetone to form acetone hydrate.
-
Using the guidelines covered in Lesson 7, identify which sentence of the three sentences is correct. All new bank customers' qualify for free checking accounts. All new bank customer's qualify for...
-
When it comes to paying state and local taxes in Texas, residents can expect to pay which of the following? Correct Answer(s) income tax Press Space to open property tax Press Space to open sales tax...
-
Which of the following forms or schedules is correct for reporting Oregon itemized deductions? Question 23Select one: a. No additional form needs to be completed, the taxpayer will need to attach...

Study smarter with the SolutionInn App


