Show syntheses of these compounds from the indicated starting materials. More than one step may b necessary.
Question:
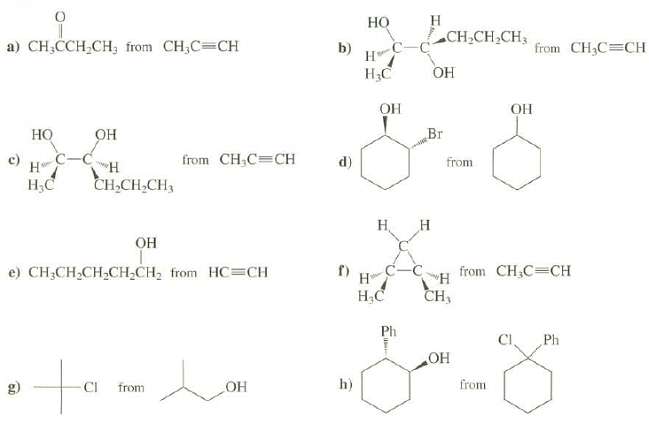
Show syntheses of these compounds from the indicated starting materials. More than one step may b necessary. Your syntheses may produce both enantiomers of any target that ischiral.
Transcribed Image Text:
Еснснсн, Но CH,CH,CH3 a) CH;CCH CH, from CH,C=CH b) from CH3C CH н НаС ОН ОН Он Br Но OH H C-C Н.С from CH,C CH c) d) from Ен,CH,CH, Н н Он from CH,C=CH e) CH;CH,CH2CH2CH2 from HC=CH н Н.С CH, Ph CI. Ph ОН to h) from g) ОН CI from
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
a CHCCH 1NaNH 2 CH3Br CHCCCH3 HgSO4 HO HSO4 b CHCCH UNaNH 2 CH3CHCHBr NaOH HO H CH ...View the full answer

Answered By

Hillary Waliaulah
As a tutor, I am that experienced with over 5 years. With this, I am capable of handling a variety of subjects.
5.00+
17+ Reviews
30+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show syntheses of these compounds from the indicated startingmaterials. a) CH,CH,CH,CH,N(CH3), from compounds with none of the CN bonds of the final product. CH3 b) CH,CH,OCHCH,CH; from alkyl halides...
-
Show syntheses of these compounds from the indication starting materials. Your syntheses may produce both enantiomers of any target that ischiral. a) CH.CH,CH,CH,CH,OCH, from CH;CH,CH,CH=CH2 CH3 b)...
-
Show syntheses of these compounds from1-bromobutane: b) TH. c)
-
from the given option three are similar to each other becausethey follow the same patter identify the option that is dissimilarto the other three A. BCED B. EFhG c. KLNM D. RSVT
-
What is the meaning of and motivation for balance in BSC?
-
If internal controls are weak, in what situations could an audit still be done, and in what situations would it not be possible to do an audit?
-
AUS. parent acquired its Singapore subsidiary at the beginning of 2017, for a price that is S\($10,000,000\) in excess of the subsidiarys book value. The excess paid was attributable to goodwill,...
-
Using the financial statements for the Snider Corporation, calculate the 13 basic ratios found in the chapter. SNIDER CORPORATION Balance Sheet December 31, 2013 Assets Current assets:...
-
Consider a 5-year default-free security with annual coupons and with a face value of $1,000. (a) What is its coupon rate if it is currently selling at par? (b) What is its coupon rate if it is...
-
To measure the capacitance of a conductor, it is first charged to a potential Vo = 1350V. It is then connected by a conducting wire to a distant metal sphere of radius r = 3cm. As a result the...
-
Show reactions that could be used to convert 1-penetene to these compounds. More than one step may bnecessary. b) CI d) . h) g)
-
This alkynes hydration reaction can occur without added Hg2+. Show all the steps in themechanism. H,SO, PhC=CH + H20 PHCCH3
-
Indicate whether each statement is true or false: (a) NaCl dissolves in water but not in benzene (C 6 H 6 ) because benzene is denser than water. (b) NaCl dissolves in water but not in benzene...
-
Explain your selected technology. What is it? How does it work? What is its purpose? Timeline: Provide a timeline of major events concerning your technology. Include major developments, regulations,...
-
Expand Your Critical Thinking Decision-Making Across the Organization CT5.1 Creative Ideas Company has decided to introduce a new product. The new product can be manu- factured by either a...
-
The following transactions occurred for the month of May. Date Units Cost Total Sales Price 1-May Beginning Balance 80 12 $960 2-May Purchase 85 15 $1,275 3-May Purchase 65 11 $715 10-May Sale 90 $30...
-
Describe three things you can do as an individual and an employee to protect the environment. What would be sustainability Initiatives you will recommend the organization you want to work for to...
-
The Wilson family are nationals of the United Kingdom (UK). George, the father, has a Bachelor of Science degree in engineering and is presently employed as a manager at Proctor and Gamble in the UK....
-
The point on the cam with maximum pressure angle is called the (a) pitch point (b) trace point (c) cam centre (d) roller centre.
-
Repeat the previous problem, but close the positions on September 20. Use the spreadsheet to find the profits for the possible stock prices on September 20. Generate a graph and use it to identify...
-
Why is H 3 O + the strongest acid and OH - the strongest base that can exist in significant amounts in aqueous solutions?
-
Draw d-ribulose in its five-membered cyclic -hemiacetalform. H- C=0 H- -HO- H Ribulose HO- H-
-
Look tip the structure of r-talose in figure, and draw the ? anomer in its pyranose form. Identify the ring sub-stituents as axial or equatorial. - CH- D-Glyceraldehyde R/L H- - -- 2R - - CH- CH-...
-
Draw structures for (he products you would expect to obtain from reaction of -D-talopyranose with each of the following reagents: (a) NaBH 4 in H 2 O (b) Warm dilute HNO 3 (c) Br 2 , H 2 O (d) CH 3...
-
Part 1: Create Car and Garage classes in accordance with the following specifications. I have provided the CarTester class as a test driver to test your Car and Garage classes. Do not change the...
-
Exception Handling CMPT220L Due on Apr 14, 2023 by 11:59 PM . Points: 100 Problems 1. Using exception handling, write a Java program that prompts the user to enter a string and displays the length of...
-
Implement a class HoneyDoList, which is used to manage a collection of Task. It should contain the following private fields: tasks[] an array of Task numTasks, a non-negative integer storing the...

Study smarter with the SolutionInn App


