Sketch what you might expect the 1H and 13C NMR spectra of the following compound to look
Question:
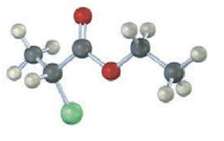
Sketch what you might expect the 1H and 13C NMR spectra of the following compound to look like (yellow-green =Cl):
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
The compound has 5 different types of ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The 1H and 13C NMR spectra of compound A, C8H9Br are shown. Propose a structure for A, and assign peaks in the spectra to your structure. TMS O ppm 10 8. 6. Chemical shift (8) TMS 200 180 160 140 120...
-
The proton and 13C NMR spectra of a compound of formula C4H11N are shown here. Determine the structure of this amine, and give peak assignments for all of the protons in the structure. 200 180 160...
-
How could you use 1H and 13C NMR to help you distinguish among the following isomeric compounds of formulaC4H8? CH2-CH2 H2H CH CCH3HCH CH2-CH2 CHH2
-
Suppose, in the New Monetarist model, that there is deficient financial liquidity. If the fiscal authority were to engineer a tax cut, financed by an increase in the quantity of government debt, with...
-
How are franchising and tapered integration similar? How do these strategies differ?
-
Israel's kibbutzim are by now well diversified beyond their agrarian roots, producing everything from lollipops to plastic pipe. These 282 widely scattered communes of several hundred members...
-
Assume that Protex Company, a U.S. company, is involved in petroleum operations in Thailand. Protex Company has a 40% WI, while the Local Oil Company has a 60% WI. Annual gross production is to be...
-
Assume Magnificent Modems, Inc. (MMI) is a division of Gilmore Business Products (GBP). GBP uses ROI as the primary measure of managerial performance. GBP has a desired return on investment (ROI) of...
-
At what point(s) on the curve x = 9t2 +7, y=t3 - 4 does the tangent line have slope. (x, y) =
-
1. Why entrepreneurship becomes so important in recent years? 2. Why are the big companies even move into Intrapreneurship?
-
How many absorptions would you expect the following compound to have in its 1H and 13C NMRspectra?
-
How many electronically nonequivalent kinds of protons and how many kinds of carbons are present in the following compound? Don?t forget that cyclohexane rings can ring-flip.
-
A function is given by a table of values, a graph, a formula, or a verbal description. Determine whether it is one-to-one.
-
Luisa Williams earns $7.50/hour, and worked 44 hours during the most recent week. She makes a 401(k) retirement plan contribution of 14% of gross pay each period. Luisa Williams is single, and claims...
-
Wildhorse Company invests $10,400,000 in 4% fixed rate corporate bonds on January 1, 2025. All the bonds are classified as available-for-sale and are purchased at par. At year-end, market interest...
-
What is the role of apoptosis in multicellular organisms, and how does it contribute to developmental processes and homeostasis ?
-
How do stem cells contribute to tissue regeneration and repair in multicellular organisms, and what are the ethical considerations surrounding their use in medical research and therapy ?
-
Kilimanjaro is the tallest mountain in Africa, and is a large,dormant volcano. Note that there is snow and glaciers at the top ofthe mountain even though it\'s very close to the equator and...
-
Loris Catering, Inc., purchased $400 of supplies on account. The journal entry to record this transaction is denoted by which of the following? DATE ACCOUNTS POST REF. DR. CR. a. Inventory 400...
-
What are the 5 Cs of marketing channel structure?
-
A solution contains 50.0 g of heptane (C 7 H 16 ) and 50.0 g of octane (C 8 H 18 ) at 25 C. The vapor pressures of pure heptane and pure octane at 25 C are 45.8 torr and 10.9 torr, respectively....
-
Predict the chemical shifts of the protons in the following compounds. (a) (b) (c) (d) (e) (f) HC(CH3)3 CH CH CH3 , - CH,CCCH OH CH,CH,-C-OH CH3 CHC-CH Br
-
Draw the NMR spectrum expected from ethanol that has been shaken with a drop of D2O.
-
Propose chemical structures consistent with the following NMR spectra and molecular formulas. In spectrum (a) Explain why the peaks around δ 1.65 and δ 3.75 are not clean...
-
The comparative statements of Oriole Company are presented here: ORIOLE COMPANY Income Statements For the Years Ended December 31 2022 2021 Net sales $1,895,640 $1,755,600 Cost of goods sold...
-
Lars Linken opened Pronghorn Cleaners on March 1, 2022. During March, the following transactions were completed. Mar Issued 9,800 shares of common stock for $14,700 cash. 1 1 1 2 3 9 6. Borrowed...
-
Outback Outfitters sells recreational equipment. One of the company's products, a small camp stove, sells for $90 per unit. Variable expenses are $63 per stove, and fixed expenses associated with the...

Study smarter with the SolutionInn App


