a. Determine the gage pressure at point A in Fig. 3.36. b. If the barometric pressure is
Question:
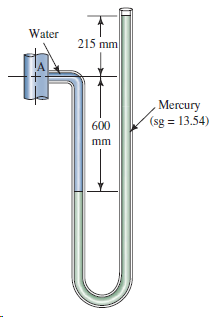
b. If the barometric pressure is 737 mm of mercury, express the pressure at point A in kPa(abs).
Transcribed Image Text:
Water 215 mm Mercury (sg = 13.54) 600 mm
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (14 reviews)
a b p atm m h 1354 981 737 ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Determine the gage pressure at point A in Fig. P2.45, in pascals, Is it higher or lower than Patmosphere? Air Oil SG-0.85 30 cm 40 cm 45 cm 15 cm Water Mercury
-
A multi fluid container is connected to a U-tube, as shown in Fig. P178, for the given specific gravities and fluid column heights, determine the gage pressure at A. Also determine the height of a...
-
An airplane is flying at an altitude of 12,000 m. Determine the gage pressure at the stagnation point on the nose of the plane if the speed of the plane is 300 km/h. How would you solve this problem...
-
Over the past 10 years, a bank has experienced the following loan losses on its C&I loans, consumer loans, and total loan portfolio. Using regression analysis on these historical loan losses, the...
-
The intangible assets section of Glover Ltd. at December 31, 2016, is presented below. Patents (60,000 cost less 6,000 amortization)...................54,000 Franchises (48,000 cost less 19,200...
-
The trial balance of Fortaleza Boutique at December 31 shows Merchandise inventory R$21,000, Sales R$136,000. Sales Returns and Allowance R$4,000. Sales Discounts R$3,000, Cost of Goods Sold...
-
Following is a TI-84 Plus display showing a 95% confidence interval for 1. a. What is the slope of the least-squares regression line? b. How many degrees of freedom are there? c. How many points are...
-
Rx Corp wants to acquire all the stock of Vit B corp. Vit B is wholly owned by Honey, an individual shareholder who has owned the stock for 5 years. Honey's basis in Vit B is $75. Vit B's assets are...
-
Find the center and radius of the sphere with the equation x2 + y2 + z2 = 6y + 18z - 2x. 9) Given a = 24i - 8j + 12k and b = 3i - j + 3/2 k , determine whether a and b are parallel, perpendicular, or...
-
Charlie is a salesman at Pick up Motors car dealership. He is performing a detailed analysis of his sales for the first half of 2018, including representing his sales and commissions with graphs....
-
Figure 3.35 shows an inclined well-type manometer in which the distance L indicates the movement of the gage fluid level as the pressure pA is applied above the well. The gage fluid has a specific...
-
What is the function of a barometer?
-
In gas adsorption a vapor is transferred from a gas mixture to the surface of a solid. (See Section 6.7.) An approximate but useful way of analyzing adsorption is to treat it simply as condensation...
-
What conditions must be in place for a battered woman syndrome defense to succeed?
-
When can an omission qualify as the actus reus of a crime?
-
When is the defense of consent not available?
-
Massachusetts is one of ten States that prohibit distribution of hypodermic needles without a prescription. In the face of those statutes the defendants operated a needle exchange program in an...
-
Explain how conspiracy differs from complicity.
-
Maintenance costs for a regenerative thermal oxidizer increased according to an arithmetic gradient for 5 years. The cost in year 1 was $7000. If the interest rate is 10% per year and the present...
-
Using Apple, demonstrate how the differentiation strategy can be well implemented.
-
Calculate q, w, U, and H if 2.25 mol of an ideal gas with C v,m = 3/2 R undergoes a reversible adiabatic expansion from an initial volume V i = 5.50 m 3 to a final volume. V f =25.0 m 3 .The initial...
-
For an ideal gas, (U/V) T and (h/P) T = 0. Prove that C V is independent of volume and C P is independent of pressure.
-
Assume the internal energy of an elastic fiber under tension (see Problem 6.16) is given by. dU = T dS P dV F dl. Obtain an expression for, (g/L) P,T and calculate the maximum non-expansion work...
-
Identify and explain the difference(s) between horizontal and vertical audits. Explain the ways that customers and stakeholders might benefit from the changes that result from conducting an audit....
-
Education is an effective tool to learn skills relating to behaviour management. Where might you access this education both internally and externally to an organisation? When would this need be...
-
Density is a physical property that relates the mass of asubstance to its volume. a) Calculate the density (in g/mL) of aliquid that has a mass of 0.155 g and a volume of 0.000275 L. a- calculate the...

Study smarter with the SolutionInn App


